Table of Contents
Atherosclerosis, also known as coronary artery disease (CAD), is the most common form of cardiovascular disease (CVD) and a leading cause of death worldwide. This condition is characterized by the buildup of lipids (fats) and inflammation in the large arteries. Over time, this accumulation can lead to severe complications like myocardial infarction (MI) and stroke. Despite a decline in prevalence in some regions, atherosclerosis remains a significant global health issue, especially among the elderly.
What is Atherosclerosis?
Atherosclerotic lesions are formed when lipids, inflammatory cells, smooth muscle cells, and necrotic cell debris accumulate in the inner lining (intima) of large arteries. This process leads to narrowing and hardening of the arteries, which can reduce blood flow. In more advanced stages, lesions may rupture, leading to blood clots that can cause heart attacks or strokes. The condition progresses slowly and is more common in older adults, though some genetic conditions can lead to early onset.
The Formation of Atherosclerotic Lesions
Atherosclerosis begins when lipoproteins, such as low-density lipoproteins (LDLs), accumulate in the intimal layer of artery walls. The damaged endothelial cells (ECs) lining the arteries attract inflammatory cells, such as monocytes, which penetrate the artery walls and transform into macrophages. These macrophages ingest oxidized lipids, forming foam cells that further contribute to plaque buildup.
As the plaque develops, the artery wall thickens, and smooth muscle cells (SMCs) migrate to form a fibrous cap over the plaque. While this helps stabilize the plaque, it can also lead to further complications, such as a ruptured plaque, which can obstruct blood flow and cause a heart attack or stroke.

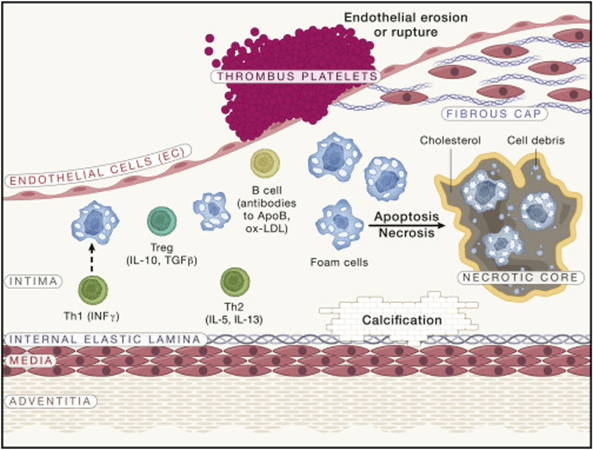
Advanced atherosclerotic lesions
Following the initial accumulation of lipids and foam cells, additional leukocytes, mainly T lymphocytes, enter the lesion and act with macrophages. The foam cells die over time, resulting in necrotic cores composed of cell debris and cholesterol. In addition, SMC change from a contractile to a proliferative state and move to the region underneath the EC to create a “fibrous cap” that shields the lesion from rupture. SMC can also develop into macrophage-like cells, which produce foam cells, and bone-like cells, which deposit calcium phosphate mineral. Although lesions can get large enough to obstruct blood flow, the most clinically relevant event is a MI caused by the development of a clot caused by lesion rupture or endothelial degradation.

Aging and Atherosclerosis
Atherosclerosis primarily affects individuals over the age of 55, though certain genetic conditions can cause early-onset disease. In older individuals, several mechanisms contribute to the progression of atherosclerosis:
- Chronic Inflammation: Low-grade inflammation is common in older adults and plays a significant role in the development of atherosclerosis.
- Clonal Hematopoiesis: This is the process where mutations accumulate in blood cells, which may lead to inflammation and an increased risk of atherosclerosis.
- Cellular Senescence: As people age, endothelial cells, smooth muscle cells, and macrophages in the arteries may undergo cellular senescence, contributing to inflammation and plaque instability.
Genetics of atherosclerosis
Recent genetic studies have identified over 200 loci associated with cardiovascular disease (CVD), helping researchers understand the hereditary factors contributing to atherosclerosis. Many of these genes affect plasma lipid levels, blood pressure, and other risk factors for atherosclerosis. Genome-wide association studies (GWAS) are providing valuable insights into genetic predispositions to heart disease.

Traditional risk factors
Several well-established factors increase the risk of developing atherosclerosis and related diseases like heart attack and stroke.
Lipoprotein metabolism:
High levels of triglyceride-rich lipoproteins (such as LDL cholesterol) are major contributors to atherosclerosis. Recent research has shown that these lipoproteins directly promote plaque formation.
Hypertension:
High blood pressure is a key risk factor for atherosclerosis. However, the exact mechanisms by which hypertension contributes to the development of CAD remain under study.
Diabetes:
Type 2 diabetes, often linked to obesity, significantly increases the risk of atherosclerosis. Diabetes is associated with metabolic changes, including insulin resistance and higher levels of LDL cholesterol.
Environmental and Lifestyle Risk Factors
Environmental and lifestyle factors can significantly affect the risk of atherosclerosis:
- Diet and Obesity: Poor diet, particularly high in saturated fats, can increase the risk of atherosclerosis by raising cholesterol and triglyceride levels.
- Exercise: Regular physical activity can lower the risk of CAD by improving glucose tolerance, reducing cholesterol, and enhancing anti-inflammatory pathways.
- Sleep and Stress: Chronic stress and poor sleep can contribute to cardiovascular disease by promoting inflammation and increasing the risk of blood clot formation.
- Smoking: Smoking doubles the risk of heart disease and stroke by promoting inflammation and damaging the arterial walls.
- Air Pollution: Long-term exposure to air pollution is linked to dyslipidemia, endothelial dysfunction, and increased risk of atherosclerosis.
Intestinal microbiota:
Bacteria in particular aid in digestion, activate the immune system, and produces special metabolites that enter the host’s blood. Microbial metabolites have emerged as important atherosclerosis mediators. Trimethylamine N-oxide (TMAO) is the most important metabolite found thus far, formed by hepatic oxidation of trimethylamine, a molecule obtained through bacterial choline and carnitine metabolism. TMAO levels are higher in those who eat a lot of choline or carnitine, or who have renal illness. TMAO tends to increase platelet reactivity and vascular inflammation, which appears to cause atherosclerosis.
The Role of Gut Microbiota in Atherosclerosis
Recent studies have shown that the gut microbiome plays a crucial role in the development of atherosclerosis. Metabolites produced by bacteria, such as trimethylamine N-oxide (TMAO), have been linked to increased inflammation and platelet reactivity, contributing to the progression of atherosclerosis.
Infections and Atherosclerosis
Bacterial and viral infections have long been linked to the development of atherosclerosis. Recent studies have highlighted an increased risk of cardiovascular disease in individuals who have recovered from infections like SARS-CoV-2, suggesting that infections can trigger or worsen atherosclerotic conditions.
Diagnosing Atherosclerosis
The diagnosis of atherosclerosis typically relies on traditional risk factors, such as cholesterol levels, blood pressure, and family history. Advances in genetic testing, including polygenic risk scores, are improving the ability to predict individual risk for heart disease and atherosclerosis at an earlier age.
Managing Atherosclerosis
The management of atherosclerosis focuses on both prevention and early intervention. Medications such as statins can lower LDL cholesterol levels and reduce the risk of heart attack. Ezetimibe works by inhibiting cholesterol absorption, while PCSK9 inhibitors help lower LDL cholesterol by improving LDL receptor function.
Lifestyle Modifications for Prevention
Making healthy lifestyle choices is critical for preventing and managing atherosclerosis:
- Dietary Changes: Plant-based diets have been shown to reduce the risk of atherosclerosis by lowering cholesterol levels and decreasing inflammation.
- Regular Exercise: Physical activity can improve overall cardiovascular health and reduce atherosclerotic risk factors.
- Stress Reduction and Sleep Hygiene: Managing stress and ensuring adequate sleep are essential for maintaining heart health and preventing atherosclerosis.
Conclusion
Atherosclerosis is a progressive condition that develops over a lifetime. Although traditional risk factors like high cholesterol, hypertension, and diabetes are well-established, lifestyle factors such as diet, exercise, and stress management play a crucial role in disease prevention. With advancements in genetic testing, diagnostic imaging, and treatment options, the outlook for individuals at risk of atherosclerosis is improving. Preventive strategies and early interventions remain essential in reducing the global burden of cardiovascular disease.
References
- Björkegren JLM, Lusis AJ. Atherosclerosis: Recent Developments. Cell. 2022 Apr 27;S0092-8674(22)00400-7. doi: 10.1016/j.cell.2022.04.004
