Table of Contents
Lactic acidosis is caused by high levels of lactic acid (lactate) and protons in the bodily secretions and is usually associated with poor clinical outcomes. The intensity and clinical setting of lactic acidosis influence its impact. Lactic acid, a carbohydrate metabolism intermediate, is mostly produced by white skeletal muscle, the brain, the skin, the renal medulla, and erythrocytes. The rate of formation in these tissues, as well as the rate of metabolism in the liver and kidneys, determines blood lactic acid concentrations.
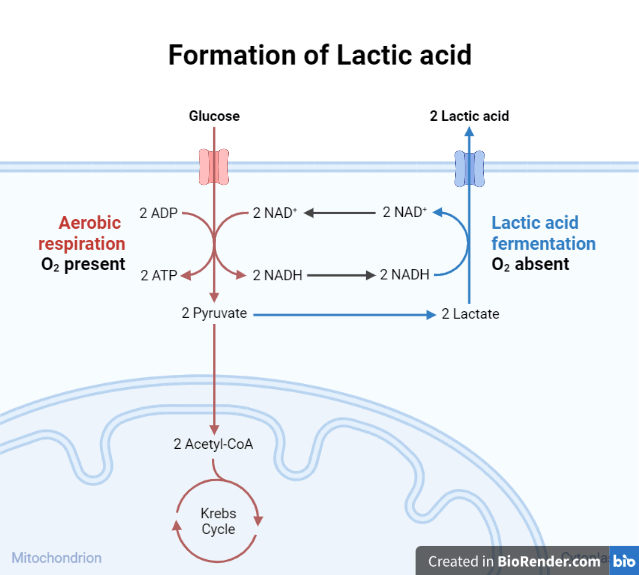
Lactic acid metabolism
Pyruvate is produced primarily by anaerobic glycolysis (E-M pathway). Lactate may be created by anaerobic glycolysis in all tissues of the body, but most tissues do not produce considerable amounts because the full oxidation of the pyruvate produced by glycolysis yields considerably more ATP. When oxygenation is insufficient, all tissues respond with an increase in lactate production. A reduction in ATP due to decreased oxidative phosphorylation permits 6-phosphofructo-1-kinase activity to rise. Under such conditions, these tissues must rely on anaerobic glycolysis for ATP synthesis, resulting in lactic acid production.
Clinical Indication of lactic acidosis
Lactic acid measurement can be used to establish and measure the severity of tissue hypoxia caused by shock or localized vascular obstruction. It is also an indicator of the overall success related to the management of those symptoms.
Hyperlactatemia:
When lactate production surpasses lactate intake, hyperlactatemia ensues. It also denotes the addition of a number of protons equal to the number of excess lactate ions, independent of the current acid–base condition. Understanding the pathophysiology of hyperlactatemia can help guide treatment. Aerobic glycolysis, which refers to accelerated glycolysis that is not dependent on tissue hypoxia, can also result in hyperlactatemia.
Causes of lactic acidosis:
Lactic acidosis occurs in two clinical outcomes:
- Type A (hypoxic), characterized by reduced tissue oxygenation and symptoms such as shock, hypovolemia, and left ventricular failure; and
- Type B (metabolic), associated with
- diseases such as (e.g., diabetes mellitus, neoplasia, liver disease),
- drugs/toxins (e.g., ethanol, methanol, salicylates), or
- inborn errors of metabolism.
Lactic acidosis is comparatively rare, affecting around 1% of hospitalized patients. It has a mortality rate of more than 60%, and it reaches 100% if hypotension is present. Type A is by far the most frequent.
Laboratory Diagnosis
Specimen:
During the blood lactic acid measurements, several precautions need to be taken. The lactic acid reference interval differs between arterial and venous blood. The usage of torniquet for a longer duration has a significant impact on the outcomes, thus it is better to collect blood torniquet free.
Normal Reference Intervals
Venous blood: 5-20 mg/dL or 0.5-2.2 mmol/L
Arterial blood: 3-7 mg/dL or 0.3 to 0.8 mmol/L
Limitations
The usage of a torniquet or clenching of the hands for a longer duration elevates lactate levels. Strenuous activity raises lactate levels in the blood. Aspirin, cyanide, ethanol, nalidixic acid, and phenoformin are drugs that raise lactic acid levels.
Clinical Significance
Increased lactic acid levels in human blood is associated with several conditions like:
- Shock
- Tissue ischemia: Anaerobic glycolysis occurs in hypoxemic organs and tissues. Lactic acid is formed as a result, resulting in elevated blood levels.
- Carbon monoxide poisoningCarbon monoxide has a stronger binding affinity to hemoglobin than oxygen. As a result, the tissues have no supply of oxygen for normal aerobic metabolism. Lactic acid is generated as a result of anaerobic metabolism, resulting in elevated blood levels.
- Severe liver disease
- Genetic errors of metabolism: Acquired and genetic abnormalities related with inadequate aerobic glucose metabolism induce increased lactic acid synthesis, which increases blood lactate levels.
- Diabetes: Lactic acid levels rise in patients with poorly managed diabetes most likely due to inefficient aerobic glucose metabolism, which causes increased lactate accumulation.
References:
- Textbook of biochemistry with clinical correlations, 4th edition by T.M. Devlin
- Medical biochemistry 5th edition by JW Baynes
- Tietz Fundamentals of Clinical chemistry and molecular diagnostics, 7th edition by C.A. Burtis
- Mosby’s manual of diagnostic and laboratory tests, 5th edition, Elsevier pub.
- Kraut, J. A., & Madias, N. E. (2014). Lactic acidosis. The New England journal of medicine, 371(24), 2309–2319. https://doi.org/10.1056/NEJMra1309483
