Table of Contents
Venipuncture is a convenient way to provide a large blood sample that can be used for many various studies. A venipuncture, also called a phlebotomy, is punctured by a hypodermic needle in the superficial vein and blood is drawn into the vacuum tube or syringe.
Performing a venipuncture involves several important steps that must be thoroughly understood before the procedure is attempted:
- Observing Standard Precautions and other safety measures throughout procedure
- Selecting the proper equipment
- Identifying the patient using two identifiers
- Preparing the patient for venipuncture
- Selecting and preparing the puncture site
- Applying and removing the tourniquet
- Obtaining the blood
- Caring for the puncture site
- Observing the patient for adverse reaction
- Labeling blood specimens immediately following blood collection
When properly performed by skilled professionals, venipuncture is a safe technique. Such personnel can include phlebotomists, health professionals, nurses, surgeons, technicians from hospitals and medical laboratory scientists. To protect the state of the vein, venipuncture must be carried out carefully. There is a lot of study and instruction under the guidance of a phlebotomist trained in the field of venipuncture to become skilled and comfortable.
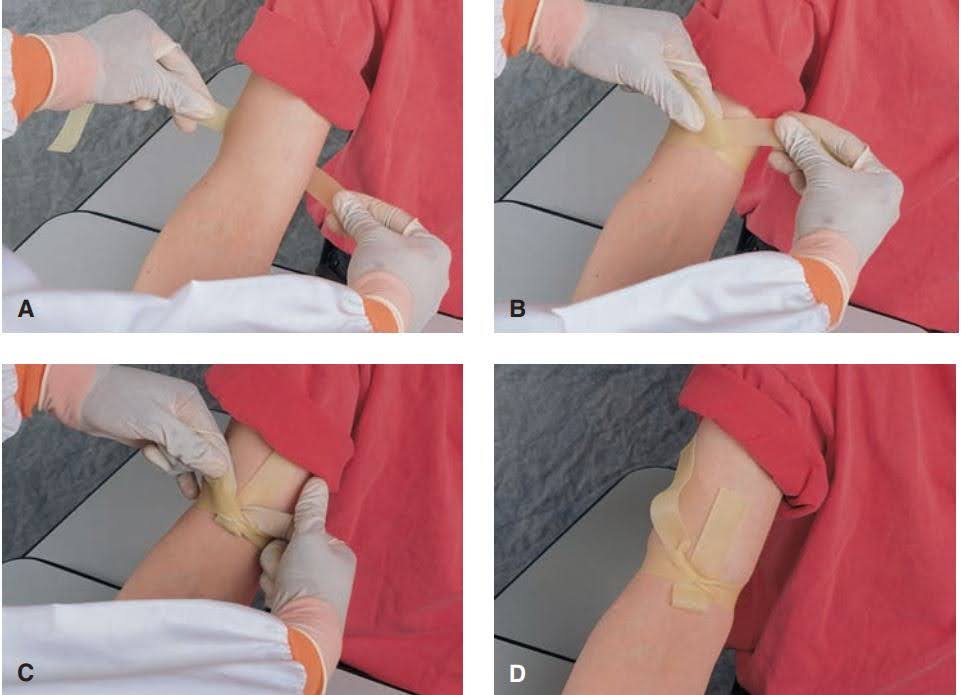
Applying a tourniquet:
- place tourniquet under arm 2-4 inches above venipuncture site;
- bring two ends of tourniquet around front of arm, stretching the two ends tight and crossing over the arm;
- maintain tension on the tourniquet ends and tuck a portion of the front tourniquet under the back forming a loop;
- the ends of the tourniquet are directed away from the venipuncture site
Non-traumatic Venipuncture:
It is essential that trauma be avoided because it may introduce tissue thromboplastin that would activate coagulation.
Order of Draw:
It is important that the correct order of draw be followed. Collect tubes containing heparin, EDTA, sodium fluoride, or clot-promoting additives before collecting tubes for coagulation research.
Specimen Processing:
Following blood processing, specimens must be collected as quickly as possible. Recommendations include processing APTT within 4 hours and PT within 24 hours. Centrifuge platelet-poor plasma to extract plasma from cells; plasma can be frozen at -20°C.
Other clotting measurements, such as the D-dimer and the PT of warfarinized subjects’ samples, are stable for 24 hours or longer.
Plasmas should be deep frozen within 4 hours of sampling unless a laboratory has evidence of the stability of measuring plasmas at room temperature for a particular test. Any clotting factor test results are steady for up to 3 months at -24 °C or lower, and up to 18 months at -74 °C (results within 10 percent of baseline defined as stable). In most cases, storing in domestic category-20°C freezers is not a viable option.
Frozen samples must be thawed quickly at 37°C for 3-5 minutes before examination. Thawing at lower temperatures is not recommended due to the risk of cryoprecipitation.
Temperature:
37°C is ideal for enzyme reactions. As temperatures rise above 37°C, labile factors V and VIII degrade. At cold temperatures, factors VII and XI are allowed.
Use Plastic or Silicone-Coated Glass Tubes:
The intrinsic pathway, which includes the contact factors prekallikrein, XI, and XII, will be activated by plain glass tubes.
Glass or plastic tubes are suitable, as long as the inside surface is non-activating, according to CLSI. It was previously thought that the use of plastic tubes in laboratories was the for a variety of reasons, including lower risk of breakage, which decreases biohazard contamination risk, and lighter weight, which lowers shipping and disposal costs.
The inner surface of the sample container used to extract blood samples may have an impact on the results (especially for screening tests) and should not be activated by touch (non-siliconized glass is inappropriate). There is evidence that findings on samples obtained in a variety of sample forms are basically synonymous for factor assays.
Anticoagulant
For coagulation specimens, 105 to 109 mmol/L (3.13 percent to 3.2 percent) tri sodium citrate (Na3C6H5O7.2H2O) with or without buffer is prescribed as an anticoagulant. This anticoagulant keeps the PH close to physiological levels and attaches calcium to avoid clotting.
Since a relative abundance of citrate from under-filling specimen tubes prolongs regular coagulation procedures, the blood-to-anticoagulant ratio is critical. The optimal blood-to-citrate anticoagulant volume ratio is 9:1.
Centrifugation
Samples should be centrifuged at room temperature (18–250C) at 150–200 g for 15 minutes to prepare platelet-rich plasma for investigation of platelet structure, and analyzed within 2 hours of sample collection. For most other tests related to bleeding disorders, samples should be centrifuged at a speed and time that produces samples with residual platelet counts below 10 ×109/L; for instance, using 2000xg speed for at least 10 minutes.
NOTE: The high hematocrit (Hematocrits >60 or <15) may result faulty PT value. Hence the blood volume and volume of anticoagulant should be corrected when high hematocrit is present. For this the volume of anticoagulant is calculated using the given below formula.
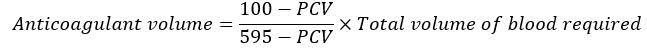
References:
- Rodak’s Hematology 5th Edition, Elsevier.
- Basic Clinical Laboratory Techniques 6th Edition, Delmar Cengage Learning
- Quality in Laboratory Hemostasis and Thrombosis, 2nd Edition
- Concise Book of Medical Laboratory Technology 2nd Edition, Jaypee
