Table of Contents
The coccidian parasite Cyclospora cayetanensis was recently identified as a human intestinal pathogen. Schneider identified it in 1881, and Ashford reported it as a human infection in 1979. Cyclosporiasis, which is caused by ingestion of sporulated oocysts in contaminated food or water, has been identified as a primary source of substantial morbidity in both immunocompetent and immunocompromised individuals.
Patients with cyclosporiasis may have gastrointestinal symptoms or be asymptomatic, and they may or may not have constitutional symptoms. Extraintestinal cyclosporiasis has also been observed, including respiratory tract infections and acalculous cholecystitis. In addition, intestinal cyclosporiasis has been linked to Reiter’s disease and Guillain–Barre syndrome.
Morphology
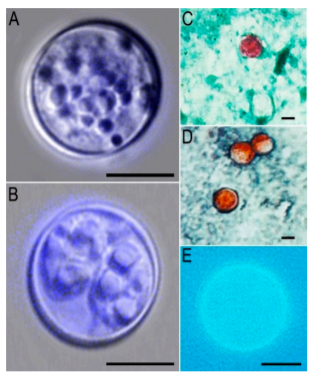
An oocyst is a morphological form seen in the feces. Two sporozoites are included in each sporocyst. As a result, each sporulated oocyst has four sporozoites. Unsporulated oocysts are spheroidal, measuring 8–10 µm in diameter and varying in size slightly. Sporulation takes place outside of the host. The wall of the oocyst is colorless, thin (1 µm), and bilayered. There is a polar body and oocyst residuum present. Sporocysts are ovoidal in shape, measuring 4 to 6 µm in length, and include both Stieda and substieda bodies, as well as a substantial residuum. A sporulated oocyst has two sporozoites in each sporocyst. The sporozoites are elongated, measuring ~1×9 µm in length. Sporozoites do not appear to have a crystalloid or refractile body.
Life Cycle
The only known host is humans. Infection occurs when a person consumes food or water contaminated with sporulated oocyst from the soil. Cyclospora cayetanensis‘ life cycle isn’t completely understood. The only stage that has been reliably recognized is the oocyst. Biopsy specimens of patients are used to determine the endogenous developmental phases.
After a host consumes sporulated oocysts from contaminated food, drink, or soil, sporozoites excyst in the gut lumen and infiltrate the enterocytes of the duodenum and jejunum epithelium, where they turn into trophozoites, which then create two forms of schizonts. 8–12 microscopic (3–4 µm long) merozoites are seen in Type I schizonts (size unknown). Four lengthy (12–15 µm long) merozoites are seen in Type II schizonts. Gamonts are formed by type II merozoites. Gamonts are unknown in terms of size. The microgamont fertilizes the macrogamont to generate the zygote during sexual multiplication.
Enterocytes produce oocysts, which are excreted unsporulated in the stool. One week is considered to constitute the prepatent period. Oocysts that have not sporulated are not infectious; they must sporulate in order to infect a host. Outside the host, sporulation will take between 7 and 14 days under laboratory conditions between 22 °C and 30 °C. Two sporocysts, each with two sporozoites, make up a sporulated oocyst.
Clinical Features
Infections with Cyclospora cayetanensis have been observed in people in over 56 countries throughout the world, spanning all five human-inhabited continents. Between June and November 1989, the first reported epidemic of Cyclospora cayetanensis infection (then known as a “alga-like organism”) occurred in Nepal among 55 British expats suffering from protracted diarrhea (https://doi.org/10.4269/ajtmh.1991.45.383).
In humans, cyclosporiasis symptoms include frequent, watery diarrhea, as well as lethargy, nausea, anorexia, cramps, and remission periods. Healthy people who have consumed sporulated oocysts often have mild-to-moderate self-limiting diarrhea. Patients with immunological deficiency, on the other hand, may suffer from severe intestine damage and diarrhea for an extended period of time. Low-grade fever and D-xylose malabsorption may also be present in certain instances. In disease-endemic areas, asymptomatic infections are also very common.
Cyclospora cayetanensis may also infect the biliary system, causing acalculous cholecystitis and the presence of oocysts in gallbladder epithelial cells in persons with acquired immunodeficiency syndrome (AIDS). Reactive arthritic disease, Reiter syndrome, and Guillain-Barré syndrome have all been linked to Cyclospora cayetanensis infection.
Laboratory Diagnosis
1. Stool examination
To rule out a Cyclospora cayetanensis infection, many stool samples should be analyzed. To obtain a detection rate of >95 percent, three stool specimens should be collected on alternate days during a 10-day period. Oocysts of Cyclospora cayetanensis can be detected by wet smears with or without iodine. For the identification of a small number of Cyclospora cayetanensis oocysts in stool samples, concentration procedures such as formalin-ether sedimentation or sucrose solution flotation can be utilized. Because of the oocysts’ tiny size (8–10 µm) and spherical form, they might be mistaken for amoebae or inflammatory cells, necessitating the employment of other microscopic approaches to improve detection sensitivity.
2. Modified Acid-Fast Staining
Acid-fast lipids are found in the walls of Cyclospora cayetanensis, Cryptosporidium, and Cystoisospora oocysts, which is a common characteristic that makes acid-fast staining useful for screening these three parasites in a single test. Although modified acid-fast staining may be used to detect these species, in Cyclospora cayetanensis, different levels of dye absorption can result in ghost cells or cells that are weakly stained with those that are well stained. To increase Cyclospora cayetanensis detection, modest modifications in modified acid-fast stains have been made. Using 1% H2SO4 as a decolorizer instead of alcohol is one of the most popular alterations. Another approach is to add dimethyl sulfoxide (DMSO) to the phenol-basic fuchsine and use acetic acid with malachite green as a combination decolorizer-counter stain to get improved penetration for observing the oocysts’ internal structures.
3. Heated Safranin Staining
The staining was first used to identify Cryptosporidium. Cyclospora cayetanensis oocysts were consistently stained a vivid reddish-orange hue using a modified heated safranin stain. Safranin staining equals the Ziehl Nielsen acid-fasting staining technique in terms of sensitivity and specificity.
3. Auto Fluorescence
Cyclospora cayetanensis, Cystoisospora, and Cryptosporidium oocysts all display autofluorescence. Cyclospora cayetanensis oocysts have a strong autofluorescence that can be used as a microscopic test for identification. When exposed to 365 nm UV light, Cyclospora cayetanensis looks blue, and when subjected to 450-490 nm UV light, it appears green. When exposed to 365 nm UV light, Cryptosporidium looks violet, and when subjected to 405-436 nm UV light, it appears green.
4. Molecular Detection and Characterization
For the diagnosis of cyclosporiasis, molecular techniques have significant advantages, including the capacity to identify different infections concurrently utilizing multiple platforms, swift analysis, and high sensitivity.
Treatment
Cotrimoxazole (trimethoprim 160 mg/sulfamethoxazole 800 mg) is used to treat cyclosporiasis and is taken twice a day for seven days. Long-term suppressive management medication may be required for HIV patients.
References
- Li J, Wang R, Chen Y, Xiao L, Zhang L. Cyclospora cayetanensis infection in humans: biological characteristics, clinical features, epidemiology, detection method and treatment. Parasitology. PMID: 31699163.
- Almeria S, Cinar HN, Dubey JP. Cyclospora cayetanensis and Cyclosporiasis: An Update. Microorganisms. 2019;7(9):317
- Hussein, E.M., El-Gayar, E.K., Ismail, O.A. et al. Identification of a Cyclospora cayetanensis Oocyst Antigens and Their Validity in the Detection of Immunogenic Patterns of Cyclosporiasis Patients. Acta Parasit. 66, 416–427 (2021).
- McHardy, I. H., Wu, M., Shimizu-Cohen, R., Couturier, M. R., & Humphries, R. M. (2014). Detection of intestinal protozoa in the clinical laboratory. Journal of clinical microbiology, 52(3), 712–720.
- Paniker’s Textbook of Medical Parasitology
- Clinical Parasitology, A practical approach.
- Essentials of Medical Parasitology
