Table of Contents
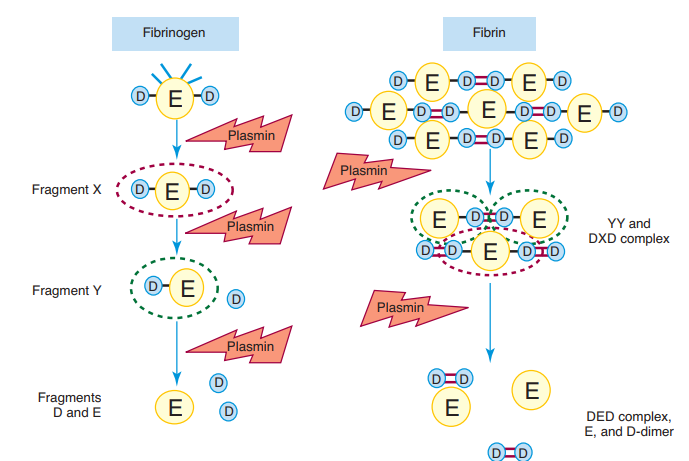
During hemostasis, coagulation system development of fibrin clots as a reaction to vascular injury is regulated by the fibrinolytic system breakup of the clot. D- Dimers are one of several fragments formed when plasmin, an enzyme that is activated by the fibrinolytic pathway, cleaves fibrin to break down clots. It consists of two covalently bound fibrin D domains which were cross-linked when the clot was formed by factor XIII. This fragment forms specific epitopes that can be targeted in D- dimer assays by monoclonal antibodies to confirm that thrombin is being produced by the coagulation cascade.
Formation of D-Dimer
Plasmin cleaves fibrin and creates a variety of identifiable fragments of the fibrin: X, Y, D , E and D-D. Several of these fragments suppress hemostasis and lead to hemorrhage by blocking platelet activation and avoiding polymerisation of the fibrin. Fragment X is defined as the central E domain with the two D domains (D-E-D), minus a few plasmin-cleaved peptides. Fragment Y is the domain E after one D domain (D-E) has been cleavage. These fragments are gradually further digested to human domains D and E.
The D-D component, called D-dimer, consists of two D domains of different fibrin molecules cross-linked by factor XIIIa ‘s action. Fragments X, Y, D , and E are formed by plasmin digestion of either fibrin or fibrinogen, but D-dimer is a particular cross-linked fibrin digestion component only and is therefore a marker of thrombosis and fibrinolysis — that is, thrombin, factor XIIIa, and plasmin activation.
Quantitative or semiquantitative immunoassay can detect the different fragments to reveal fibrinolytic reaction. D-dimer is detected independently by monoclonal antibody for D-dimer antigen using a broad range of standardized validated laboratory immunoassays and other methods including complete blood point-of – care studies. The D-dimer immunoassay is used to classify persistent and severe DIC in reported cases of deep venous thrombosis or pulmonary embolism, and to rule out venous thromboembolism.
D-Dimer Assays
D-dimer assays utilize mono- or polyspecific D-dimer antibodies to provide quantitative or qualitative evidence on the D-dimer content of whole blood or plasma. D-dimer is the result of cross-linked fibrin lysis, and D-dimer levels are elevated in acute Venous Thrmboembolism (VTE) patients. However, the examination is unspecific and in a number of other circumstances, including malignancy, autoimmune diseases, and pathogens, the amount of D-dimer can be elevated. Hence the D-dimer test is particularly effective as a method for removing alleged DVT.
Principles of D-Dimer measurement
The process of measuring D-dimer can be conceptually divided into two steps:
- D-dimer fragments must be monoclonal antibodies
- D-dimer plus monoclonal antibodies must be detected and quantified
Captured antibodies are often immobilized on a wider system, such as a well or membrane microplate, or attached to a latex bead or red cell. Antibodies for identification may be labelled and create a colorimetric or fluorescent reaction that is quantified after binding captured D- dimer or can be bound to a large object such as a bead or red blood cell for agglutination purposes. In sandwich-based assays, the D-dimer molecule may provide specificity or various epitopes on the capture and detection antibodies. It follows that the two separate epitopes found in a D- dimer molecule must be identified by such assays. In agglutination-based assays, the same monoclonal antibody acts as both the capture and detection antibody. These assays grab only fragments of D- dimer with several similar epitopes.
Conditions characterized by increased D-Dimer concentrations
| Old age | Stroke |
| Neonatal period | Peripheral arteriopathy |
| Pregnancy | Aneurism |
| Hospitalization | Congestive cardiac failure |
| Disability | Hemolysis (falciform anemia) |
| Infection | Hemorrhage |
| Tumor | Acute respiratory distress syndrome |
| Recent surgery | Liver or renal disease |
| Trauma, burns | Inflammatory bowel disease |
| DIC | Thrombolytic therapy |
| VTE | Aortic dissection |
| Ischemic cardiopathy |
Limitation of the D-Dimer Assay
D-dimer assays have two principal limitations:
- A positive test result is nonspecific and should not be used as the sole criterion for diagnosis of VTE.
- There are various test kits available that have varying VTE sensitivities.
But the effects of the D-dimer cannot be interchanged between kits. D-dimer assays follow varying methods with some utilizing fibrinogen, while some follow D-dimer. This results in variations in documentation as cut-offs from labs rely on which norm is being used. This has contributed to physicians raising concern in utilizing D-dimer assays. In addition, the use of an insensitive D-dimer assay to rule out VTE may contribute to the absence of needed diagnostic tests, thereby putting patients at risk for PE and death.
