Table of Contents
Hepatic Lipase belongs to the lipase gene family, which also includes pancreatic lipase, Hepatic Lipase, and endothelial lipase. In humans, the Hepatic Lipase (HL) gene is found on chromosome 15 (q15–q22). It is more over 60 kb in length, with eight introns and nine exons accounting for 1.6 kb. Hepatic Lipase is a 65-kD glycoprotein that is largely generated and released by hepatocytes and, to a lesser degree, macrophages. HL may hydrolyze triglycerides (TG) and phospholipids (PL) in a variety of lipoproteins, but it is most prominent in the conversion of IDLs to LDLs and the conversion of post-prandial triglyceride-rich HDL to post-absorptive TG low HDL. The traditional notion of HL’s function in plasma lipid and lipoprotein metabolism.
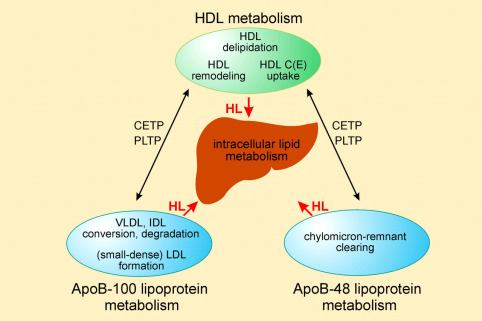
Metabolism
Hepatic Lipase hydrolyzes both phospholipids (PL) and triglycerides (TG). It primarily hydrolyzes phosphatidylethanolamine and TG containing long chain unsaturated fatty acids. It is released from the surface of hepatic nonparenchymal cells by intravenous heparin injection and is commonly measured in humans as enzyme activity in “post-heparin” plasma (PHP). Heparin injection also causes lipoprotein lipase (LPL) to be released into the plasma. Hepatic Lipase differs from LPL in that it is not inactivated by 1 M NaCl or protamine sulfate, and it is not activated by apolipoprotein CII. In vitro, apolipoproteins have been shown to both inhibit and stimulate HL. Hepatocytes produce and secrete hepatic lipase, which was previously assumed to be bound to the surface of liver endothelial cells. However, a reevaluation has revealed that HL is predominantly attached to glycosaminoglycans on the surface of hepatocytes.
Polymorphism of HL gene
According to research, the LIPC promoter -514C>T polymorphism is related with a modest reduction in HL activity and an increase in HDL-C levels. According to one research, HL activity in post-heparin plasma (PHP) was 15–20% lower in heterozygotes and 30% lower in homozygotes for the -514T gene in males with coronary artery disease (CAD). Hokanson and colleagues discovered a link between the LIPC-480C>T polymorphism (the same as the -514T allele) and subclinical CAD in type 1 diabetes. According to these findings, people who have a low HL gene are more likely to develop atherosclerosis.
Prevalence of HL Deficiency
Hepatic Lipase deficienccy is one of the rarest disorders of lipoprotein metabolism, with just few cases recorded. One of these cases had mixed HL and LPL insufficiency, which likely obscures the clinical and biochemical characteristics that are distinctive to isolated HL deficiency. This individual has been discovered as a homozygote for a mutation in the DNA sequence that impairs the normal splicing of the gene.
Hepatic Lipase Estimation
To determine whether HL is clinically pro- or anti-atherogenic, it would be preferable if HL could be measured in serum without the use of heparin. HL protein mass is currently only measurable in post-heparin plasma (PHP) as a material, whereas LPL protein mass is measurable in both serum and post-heparin plasma (PHP).
The amount of oleic acid produced by the enzyme-catalyzed hydrolysis of an emulsion containing [3H]-triolein can be used to determine hepatic lipase activity in PHP. The assay mixture contains labelled and unlabeled triolein, 0.11 mmol/ml of L-lysophosphatidylcholine, and 0.2 percent bovine serum albumin in 0.2 M buffer (Tris–HCl pH 8.8 with NaCl 0.15 M). This mixture is incubated for 30 minutes at 30 0C with post-heparin plasma (PHP) in saline solution 1: 10 and NaCl 1 M (as an inhibitor of the other lipases).
After incubation, the reaction was stopped, and the released fatty acids are isolated using a carbonate-borate buffer with a pH of 10.5. The [3H] oleic acid is quantified using a Liquid Scintillation Analyzer. The difference in counts per minute between the blank and sample is used to calculate hepatic lipase activity. The results are expressed as mmol free fatty acids/h/ml post-heparin plasma (PHP), with CV intra-assay at 4% and CV inter-assay at 9%.
Endothelial Lipase and Hepatic Lipase
It was discovered that plasma endothelial lipase (EL) activity was inversely related to HDL-C levels, and that EL activity in CAD patients was significantly higher than in non-CAD patients. Several studies have found that EL levels are elevated in people with metabolic syndrome. Furthermore, it has been discovered that inflammation is linked to an increase in EL. In relation to this, it has been reported that there is a weak but significant inverse correlation between EL and adiponectin levels. However, studies, including our own, show that there are inverse correlations between adiponectin levels and HL activity. Furthermore, it is known that HL activity is increased in metabolic syndrome or insulin resistance states. These previous findings suggest that changes in EL and HL in metabolic disorders are similar.
Treatment
There were reductions in plasma cholesterol by lovastatin and those in TG by gemfibrozil.
References
- Miksztowicz V, Lucero D, Zago V, Cacciagiú L, Lopez G, Gonzalez Ballerga E, Sordá J, Fassio E, Schreier L, Berg G. Hepatic lipase activity is increased in non-alcoholic fatty liver disease beyond insulin resistance. Diabetes Metab Res Rev. 2012 Sep;28(6):535-41. doi: 10.1002/dmrr.2312. PMID: 22539458.
- Kobayashi J, Miyashita K, Nakajima K, Mabuchi H. Hepatic Lipase: a Comprehensive View of its Role on Plasma Lipid and Lipoprotein Metabolism. J Atheroscler Thromb. 2015;22(10):1001-11. doi: 10.5551/jat.31617. Epub 2015 Jul 21. PMID: 26194979.
- Connelly PW, Hegele RA. Hepatic lipase deficiency. Crit Rev Clin Lab Sci. 1998 Dec;35(6):547-72. doi: 10.1080/10408369891234273. PMID: 9885775.
