Table of Contents
Low-density lipoprotein cholesterol, or LDL cholesterol, is a kind of fat that circulates in the blood, transporting cholesterol across the body to where it’s needed for cell repair and depositing it inside artery walls. Because cholesterol and triglycerides are water insoluble, they must be bound to proteins in order to pass through the hydrophilic bloodstream.
The surface membrane of the LDL particle is composed of unesterified cholesterol, whereas the hydrophobic interior is composed of fatty acid esters of cholesterol. The membrane contains one copy of the hydrophobic apo-B protein, which mediates LDL particle binding to particular cell-surface receptors.
LDL particles are the primary transporters of cholesterol in the human circulatory system, and they play an important role in cholesterol transfer and metabolism. LDL is a diverse collection of particles with a wide range of size, content, and structure, with a density of 1.019 to 1.063 kilograms per liter. A hydrophobic core of nonpolar triglyceride and cholesteryl ester molecules is surrounded by a monolayer of phospholipids (PL), mostly phosphatidylcholine and sphingomyelin, in LDL particles.

Mechanism of LDL formation
The liver and most other organs have the LDL receptor. It detects Apo B and Apo E and uses endocytosis to transport LDL, chylomicron remnants, and Intermediate density lipoprotein (IDL). The lipoprotein particle is destroyed in lysosomes after internalization, and cholesterol is liberated. HMG CoA reductase activity rises when cholesterol enters the cell, which subsequently synthesizes cholesterol and regulates the expression of LDL receptors. Plasma LDL levels are determined by LDL receptors in the liver. When the number of receptors in the body is minimal, the liver can only take up so much LDL from the blood, resulting in high plasma LDL levels. When there are more LDL receptors, the liver absorbs more LDL from the blood, resulting in lower plasma LDL levels.
The amount of LDL receptors in a cell is controlled by cholesterol levels. When a cell detects a drop in cholesterol levels, the transcription factor Sterol regulatory-element binding proteins (SREBP) is transferred from the endoplasmic reticulum to the Golgi, where proteases cleave and activate SREBP, causing it to travel to the nucleus and enhance LDL receptor expression. When a cell’s cholesterol levels are low, high SREBP levels stay inactive in the endoplasmic reticulum, and LDL receptor expression is reduced.
Clinical significance
LDL cholesterol (LDL-C) and apoB levels in the blood are linked to the risk of atherosclerosis. The extracellular buildup of LDL-derived lipids in the form of tiny lipid droplets and vesicles, which can contribute to the formation of atherosclerotic lesions in the artery intima, is a hallmark aspect of early atherogenesis.
LDL cholesterol levels are linked to an increased risk of cardiovascular disease. Diabetes, hypertension, hypertriglyceridemia, and atherosclerosis are all frequent symptoms. Hypercholesterolemia, which is usually asymptomatic, may be associated with metabolic syndrome, resulting in hypertension in the patient. Patients with more severe hypercholesterolemia may have xanthomas on the Achilles tendon with yellow nodules or plaques, such as xanthelasma on the eyelids or corneal arcus white rings bordering the cornea.
Normal Serum Levels
Total cholesterol levels of more than 200 mg/dL and LDL cholesterol levels of more than 130 mg/dL are deemed abnormal in the fasting lipid panel.
Measurement of LDL Cholesterol
The ultracentrifugation-polyanion precipitation / Beta Quantification (ßQ) method for measuring LDL-C is not appropriate for regular laboratory testing since it is a costly, tedious, difficult, and time-consuming procedure that also necessitates high sample quantities. Below are some of the most frequent LDL cholesterol measurement methods (direct and calculated).
β-Quantification
For at least 18 hours at 105,000g, an aliquot of plasma is ultracentrifuged at density 1.006 kg/L. If chylomicrons and -VLDL are present, VLDL accumulates as a floating layer (top fraction). LDL, HDL, and any IDL and Lp(a) are mostly found in the infranatant (bottom fraction). The top fraction is removed, and the bottom fraction is quantitatively collected, reconstituted to a known volume, and the cholesterol determined. The difference between the bottom fractions of d 1.006 and HDL-C is used to compute LDLC: [d 1.006 kg/L bottom-C] LDL-C = [d 1.006 kg/L] –HDL-C.
Friedewald equation
The Friedewald calculation is built on the hypothesis that total cholesterol is distributed among the three major lipoprotein classes (HDL, LDL, and VLDL), and that VLDL carries the majority of circulating TGs, so VLDL-cholesterol (VLDL-C) can be estimated reasonably well from measured total TGs (TG/5 for mg/L or TG/2.2 for mmol/L units). The following formula is used to calculate LDL-C:

The LDL-C calculated using the equation also includes the concentrations of IDL-C and Lp(a)-C, similar to β-quantification. In clinical laboratories and large-scale investigations, this approach is the most often adopted.
The calculation’s well-known shortcomings mostly involve three instances in which the equation cannot be used:
- When chylomicrons are present, they contain proportionally less cholesterol relative to triglycerides than VLDL, resulting in an overestimation of VLDL-C and an underestimate of LDL-C. A fasting specimen (8–12 hours) is necessary since postprandial specimens frequently contain evidence of chylomicrons.
- The buildup of residual lipoproteins with an elevated proportion of cholesterol relative to triglycerides in individuals with Type III hyperlipoproteinimia or dysbetalipoproteinemia, resulting to miscalculation of VLDL-C and overestimation of LDL-C.
- When triglyceride levels increases, the proportion of cholesterol to triglycerides in VLDL lowers, resulting inaccuracies.
As a result, only specimens with triglyceride levels less than 400 mg/dL should be calculated. However, even at triglyceride values of 200 to 400 mg/dL, its reliability is significantly reduced. Specimens with triglyceride concentrations below 200 mg/dL have the best agreement with β -quantification (86–92 percent of the samples show deviations of less than 10%), whereas specimens with triglyceride concentrations of 200–300 mg/dL and 300–400 mg/dL have only 75 and 61 percent of the samples show deviations of less than 10%, respectively.
Martin-Hopkin Equation
Martin et al. suggested a unique technique of adding an adjustable factor to the triglycerides/VLDL-C ratio based on triglyceride and non-HDL-C concentrations to overcome various limitations of the Friedewald equation. Martin’s technique was calculated by taking the median of the triglyceride-to-VLDL-C ratios by non-HDL-C and triglyceride stratum among 900 605 individuals.
Rather of assuming a constant factor of 5, it uses an adjustable factor depending on triglyceride and non–HDL-C concentrations to calculate the TG:VLDL-C ratio. The 180-cell method may be programmed into an internet calculator, smartphone app, or automated laboratory reporting system. The greatest benefit comes when LDL-C values are less than 70 mg/dL, especially in individuals with high triglyceride levels. Aside from the innovative analytic technique, one of the study’s primary strengths is its size, which is 3015 times larger than the original Friedewald database.
The Martin equation is similar to the Friedewald equation in that it divides TG by an adjustable factor that may be found in a 180-cell table that displays the optimal factor based on the plasma TG and nonHDL-C levels database.
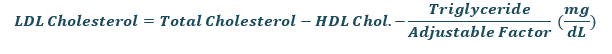

The Martin/Hopkins formula, introduced in 2013, has been shown to improve the accuracy of LDL-C estimation under a variety of conditions; it has been globally validated and is recommended for use by professional consensus groups, especially in cases of very low LDL-C, non-fasting samples, and mild hypertriglyceridemia, where the Friedewald equation is less accurate.
However, it should be emphasized that using the Martin/Hopkins method is more difficult and necessitates integrating the formula’s 180-cell approach into automated laboratory information systems or using a smartphone application for each patient. In addition, instead of indirect estimate, a direct LDL-C test should be performed in situations of moderate or severe hypertriglyceridemia.
Sampson LDL Equation
In hypertriglyceridemia patients (with triglycerides up to 800 mg/dL), this equation agreed better with β -quantification VLDL-C results than the Friedewald and Martin-Hopkins equations. Based on the β -quantification LDL-C data, an optimized LDL-C equation was created, which contained coefficients for each term as well as an intercept to optimize the fit (total cholesterol [shown as TC] and triglycerides [shown as TG]; all in mg/dL):

In the event of hypertriglyceridemia and decreasing LDL-C for treatment reassessment, Sampson’s technique offers an enticing alternative to estimate VLDL-C and LDL-C in the context of lipid profile variations. This provides for a more comprehensive assessment of cardiovascular risk management in with familial combined hyperlipidemia (FCHL) by increasing VLDL-C prediction, the most variable component in LDL-C estimate, and perhaps allowing for more accurate estimation of residual cholesterol.
Vargas-Vázquez et al., 2021 demonstrated that Sampson’s equation estimates VLDL-C and LDL-C better than Friedewald’s and Martin’s formulas, with significantly higher correlation and agreement with VLDL-C measured by ultracentrifugation and LDLC estimated using these VLDL-C measures in subjects with familial combined hyperlipidemia (FCHL).
Free Online LDL Calculator – Friedewald, Martin-Hopkins & Sampson FormulasLDL Calculator
References:
- Pirahanchi Y, Sinawe H, Dimri M. Biochemistry, LDL Cholesterol. [Updated 2021 Aug 15]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK519561/
- Bairaktari ET, Seferiadis KI, Elisaf MS. Evaluation of methods for the measurement of low-density lipoprotein cholesterol. J Cardiovasc Pharmacol Ther. 2005 Mar;10(1):45-54. doi: 10.1177/107424840501000106. PMID: 15821838.
- Wolska A, Remaley AT. Measuring LDL-cholesterol: what is the best way to do it? Curr Opin Cardiol. 2020 Jul;35(4):405-411. doi: 10.1097/HCO.0000000000000740. PMID: 32412961; PMCID: PMC7360339.
- Karkhaneh A, Bagherieh M, Sadeghi S, Kheirollahi A. Evaluation of eight formulas for LDL-C estimation in Iranian subjects with different metabolic health statuses. Lipids Health Dis. 2019 Dec 28;18(1):231. doi: 10.1186/s12944-019-1178-1. PMID: 31883533; PMCID: PMC6935216.
- Christopher D Koch, Joe M El-Khoury, New Sampson Low-Density Lipoprotein Equation: Better Than Friedewald and Martin-Hopkins, Clinical Chemistry, Volume 66, Issue 8, August 2020, Pages 1120–1121, doi.org/10.1093/clinchem/hvaa126
- Martin SS, Blaha MJ, Elshazly MB, Toth PP, Kwiterovich PO, Blumenthal RS, Jones SR. Comparison of a novel method vs the Friedewald equation for estimating low-density lipoprotein cholesterol levels from the standard lipid profile. JAMA. 2013 Nov 20;310(19):2061-8. doi: 10.1001/jama.2013.280532. PMID: 24240933; PMCID: PMC4226221.
- Piani F, Cicero AFG, Borghi C, D’Addato S; BLIP Study group. Is the 2020 Sampson equation the best formula for LDL-C estimation? Eur J Intern Med. 2021 Jan;83:99-101. doi: 10.1016/j.ejim.2020.09.009. Epub 2020 Sep 22. PMID: 32978038.
- Vargas-Vázquez A, Bello-Chavolla OY, Antonio-Villa NE, Mehta R, Cruz-Bautista I, Aguilar-Salinas CA. Comparative assessment of LDL-C and VLDL-C estimation in familial combined hyperlipidemia using Sampson’s, Martin’s and Friedewald’s equations. Lipids Health Dis. 2021 May 5;20(1):46. doi: 10.1186/s12944-021-01471-3. PMID: 33952259; PMCID: PMC8101115.

Thank you for the table. But nowadays only the USA and Liberia use imperial units whereas the remaining about 7,6 billion inhabitants of the planet use SI-units. Therefore, the table would be more appreciated and used it it was given in mmol/L