Pelger-Huët anomaly (PHA), also known as true or congenital PHA, is an autosomal dominant condition marked by reduced nuclear segmentation (bilobed, unilobed) and a distinctive coarse chromatin clumping pattern that can involve any leukocyte, while morphologic differences are more noticeable in mature neutrophils. A mutation in the lamin β-receptor gene causes the Pelger-Huët anomaly. The lamin receptor is an inner nuclear membrane protein that integrates β-type lamins and heterochromatin and plays a role in leukocyte nuclear changes.
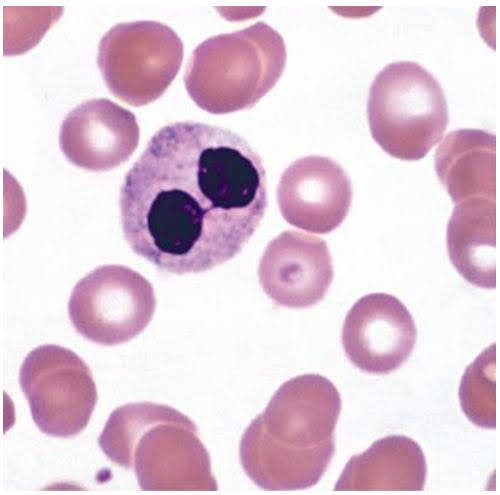
Although the precise pathological pathways remain unknown, mutations in the lamin b-receptor gene cause the morphologic changes that are typical of PHA. Round, ovoid, or peanut-shaped nuclei are all possible. Bilobed types have the distinctive spectacle-like (“pince-nez”) morphology, with nuclei connected by a thin filament.
In homozygous PHA, all neutrophils are affected and demonstrate round nuclei, whereas in the heterozygote, 55% to 93% of the neutrophil population are affected, and there is generally a mixture of all of the aforementioned nuclear shapes. Neutrophils in Pelger-Huët anomaly appear to function normally.
Pseudo- or Acquired Pelger-Huët Anomaly
Patients with hematologic malignancies such as myelodysplastic syndromes (MDS), acute myeloid leukemia, and chronic myeloproliferative neoplasms can have neutrophils with PHA morphology. Pseudo-PHA neutrophils can also be used in HIV patients, tuberculosis patients, Mycoplasma pneumoniae patients, and patients with significant bacterial infections. Mycophenolate mofetil, valproate, sulfisoxazole, ganciclovir, ibuprofen, and chemotherapies like paclitaxel and docetaxel have all been known to trigger pseudo-PHA.
Laboratory Issues in Pelger-Huët Anomaly (True/Congenital and Pseudo/Acquired)
It’s difficult to tell the difference between real PHA and pseudo-PHA.
- The number of cells with PHA morphology is an important factor to remember. The number of cells affected in real PHA is significantly greater than in pseudo-PHA (63 percent to 93 percent vs. ,38 percent , respectively).
- In true PHA, the nuclear structure and chromatin composition of all WBC lineages are significantly impaired.
- Except in some cases of MDS, where monocytes, eosinophils, and basophils can display PHA morphology, pseudo-PHA is normally only seen in neutrophils.
- Furthermore, if true PHA is assumed, a thorough analysis of family members’ peripheral blood smears can show identical results.
- In MDS-related pseudo-PHA, hypogranular neutrophils are a popular finding. Neutrophils with true PHA have natural granulation.
Difference Table: Real PHA vs. Pseudo-PHA
| Characteristics | Real PHA | Pseudo-PHA |
|---|---|---|
| Number of affected cells | Significantly greater (63% to 93%) | Relatively fewer (38%) |
| Nuclear structure and chromatin composition | Impaired in all WBC lineages | Not significantly impaired, except in some cases of MDS |
| Types of affected cells | All WBC lineages | Mainly neutrophils |
| Peripheral blood smear analysis | Identical results in family members’ smears | Results may vary among family members |
| Neutrophil characteristics | Natural granulation in neutrophils | Hypogranular neutrophils |
The clinical lab faces challenges of cell recognition in both real and pseudo-PHA. That the Pince-nez is made up of two rounded segments that are joined together by a filament. Pelger-Huët neutrophil nuclei may look circular, oval, or peanut shaped, and the cells may be identified and counted as myelocytes, metamyelocytes, or band neutrophils, mimicking a neutrophilic left shift and causing a clinical workup to determine the cause. Pelger-Huët cells, which are adult, and neutrophils, which are less mature, can be distinguished by examining the chromatin structure. Immature neutrophils, including metamyelocytes and myelocytes, may also have certain cytoplasmic basophilia.
Except for the color imparted by natural cytoplasmic granulation, the cytoplasm of mature PHA cells is almost colorless. Another lab challenge is deciding the best Pelger-Huët cell marker. Since PHA neutrophils may be unilobed or bilobed, the term “segmented neutrophil” doesn’t seem to suit. For the same purpose, the term “band neutrophil” is inappropriate. Both morphologic forms of PHA neutrophils should be labeled with the same mark. Laboratories should solve this issue by developing uniform labeling for all morphologic forms of PHA, with the aim of ensuring that the clinician knows that PHA cells are present and that lineage maturity has not been left transferred. Count Pelger-Huët neutrophils as “others,” then describe “others” as Pelger-Huët neutrophils.
References:
- Bernadette F. Rodak. George A. Fritsma, Kathryn Doig. (2007). Hematology : clinical principles and applications. St. Louis, MO :Saunders Elsevier.
