A spectrophotometer is an analytical instrument that measures analytes in a solution in both qualitative and quantitative ways. It is the most flexible, reliable, and commonly used instrument in clinical chemistry for clinical diagnosis of blood, urine, or tissues. The majority of clinical chemistry procedures have been designed to create an end product (color) that can be detected and measured using a spectrophotometer of some kind. A spectrophotometer is used as a final readout device even in highly automated analyser systems. An analyte is detected and quantified using a visible (400-800 nm) or ultraviolet wavelength in the Spectrophotometric process (below 380 nm).
Basics
Over a certain wavelength range, every chemical compound absorbs, transmits, or reflects light (electromagnetic radiation). The absorption of electromagnetic radiation in the visible, ultraviolet (UV), and infrared (IR) ranges is the basis for spectrophotometry. The electromagnetic spectrum includes everything from ultra-short wavelengths like X-rays to ultra-long wavelengths like microwaves and transmitted radio waves. All of these radiations of the same nature and move at the speed of light. They vary from one another in terms of frequency and wavelength, as well as the influence they have on matter interaction.
Light acts as if it is made up of discrete energy packets called photons, whose energy is inversely proportional to the wavelength, in addition to having wavelength characteristics. For example, ultraviolet (UV) radiation with a wavelength of 200 nm has more energy than infrared (IR) radiation with a wavelength of 750 nm. Spectrophotometry can be divided into two different categories depending on the wavelength spectrum of the light source:
- UV–visible spectrophotometer: Uses light over the UV range (185–400 nm) and visible range (400–700 nm) of electromagnetic radiation spectrum.
- IR spectrophotometer: Uses light over the IR range (700–15 000 nm) of electromagnetic radiation spectrum.
The most common spectrophotometers are used in the UV and visible regions of the spectrum, and some of these instruments also operate into the near‐IR region as well.
Relationship between Transmittance and Absorbance
When an incident light beam of intensity I0 passes through a square cell containing a solution of a compound that absorbs light of a specific wavelength and the transmitted light beam’s intensity is I, the light transmittance is defined as:
T=I/I0 —————–(1)
However, some of the incident light can be reflected by the cell’s surface or absorbed by the cell wall or the solvent. These factors are removed by using a reference cell that is similar to the sample cell except for the absence of the compound of interest from the reference cell’s solvent. The transmittance (T) through this reference cell is equal to IR divided by I0, and the transmittance for the compound in solution is equal to I divided by IR. In practice, the reference cell is inserted, and the instrument is set to an arbitrary scale reading of 100 (equivalent to 100 percent transmittance), after which the percent transmittance reading on the sample is taken. The amount of light absorbed (A) as the incident light passes through the sample is equivalent to:
A= -logI/IR —————(2)
| Wavelength, nm | Region Name | Observed |
| <380 | Ultraviolet | Invisible |
| 380-440 | Visible | Violet |
| 440-500 | Visible | Blue |
| 500-580 | Visible | Green |
| 580-600 | Visible | Yellow |
| 600-620 | Visible | Orange |
| 620-750 | Visible | Red |
| 750-2500 | Near Infrared | Not visible |
| 2500-15000 | Mid Infrared | Not visible |
| 15000-1000000 | Far Infrared | Not visible |
Beer’s Law
Beer’s Law is used in spectrophotometer measurements (sometimes referred to as the Beer-Lambert Law). According to Beer’s law, a substance’s concentration is equal to the amount of light absorbed or inversely proportional to the logarithm of the transmitted light. Beer’s rule is written mathematically as
A = abc ————(3)
Where,
A = absorbance;
a = proportionality constant defined as absorptivity;
b = light path in centimeters; and
c = concentration of the absorbing compound, usually expressed in grams per liter.
This equation is the foundation of quantitative absorption photometry research. Since absorbance (A) values do not have units, the units for ‘a’ are the equivalent of the units for ‘b’ and ‘c’. When b is 1 cm and c is expressed in moles per liter, the constant ‘a’ is replaced by the symbol epsilon (ε). The molar absorptivity (ε) is a constant for a given compound at a given wavelength under defined solvent, temperature, pH, and other conditions.
Application of Beer’s Law
In practice, the direct proportionality between absorbance and concentration must be determined experimentally for a particular instrument under specific conditions. A linear association is also seen up to a certain concentration or absorbance. When this happens, the solution is said to follow Beer’s rule up until this point. Under this constraint, a calibration constant (K) can be measured and used to measure the concentration of an unknown solution using a calibrating solution. From above Equation (3)
a = A/bc ————————-(4)
Therefore,
A1/b1c1 = A2b2c2 —————–(5)
Where, subscripts 1 and 2 indicate the absorbance (A), path length (b), and concentration (c) of calibrating and unknown solutions, respectively.
Because the light path (b) remains constant in a given method of analysis with a fixed cuvet size, b1 = b2, Equation (5) then becomes
A1/C1 = A2/C2 or Ac/Cc=Au/Cu ————-(6)
Where c and u represent calibrator and unknown, respectively.
Solving for the concentration of unknown,
Cu = Au /Ac ×Cc ————–(7)
Or
Cu = Au × Cc/Ac
=Au × K ——————————(8)
Where K = Cc/AC. The value of the constant K is obtained through measurement of the absorbance (AC) of a calibrator of known concentration (CC).
Limitations of Beer’s Law
When using calibration constants, certain measures must be taken. When the calibrator or uncertain readings surpass the linear portion of the calibration curve (i.e. when the curve no longer obeys Beer’s law), the constant should not be used. In order to construct a calibration curve, at least two, and ideally more, calibrators must be used. If a large number of calibrators with different concentrations are used, a nonlinear calibration curve may be used to cover the entire range of readings on unknowns.
Beer’s law is followed only if the following conditions are met:
- The incident light is monochromatic on the subject of study.
- The solvent absorption is insignificant, compared with the solute absorbance.
- The solute concentration is within given limits.
- An optical interferant is not present.
- A chemical reaction does not occur between the molecule of interest and another solute or solvent molecule.
Measurement without reference material of known concentration
In certain cases, a pure reference material may not be readily available, so constants derived from pure materials and recorded in the literature may be used instead. In general, published constants can only be used if the procedure is meticulously followed and measurements are performed on a spectrophotometer capable of producing high-purity light at a known wavelength. The use of broader-band light sources usually results in a reduction in absorbance.
For example, the molar absorptivity of reduced nicotinamide adenine dinucleotide (NADH) at 340 nm is frequently used as a means of determining enzyme activity. This value is only permissible under the carefully regulated conditions mentioned previously, and it should not be used until these conditions are met. Published molar absorptivities and absorption coefficients can only be used as guidance before readings on pure reference materials for a given instrument confirm them
For instance without reference the enzyme activity of ALT/ALT is calculated using theoretical factor:
ALT/AST Activity =Delta Absorbance × Factor
Where, Factor is obtained as:
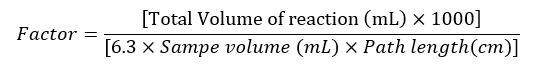
During calculation of factor, 6.3 is the Molar extinction coefficient of NADH at 370C and 340 nm wavelength.
Note: The molar extinction coefficient differs in accordance with temperature and wavelength.
Instrumentation
A spectrophotometer is a more sophisticated type of a colorimeter. Filters are used in a colorimeter to allow a wide range of wavelengths to pass through, whereas a prism or grating is used in a spectrophotometer to separate the incident beam into different wavelengths. As a result, a spectrophotometer is more effective and compact than simple colorimeters at isolating monochromatic radiation. The light from the source is broken into a parallel beam and sent to a monochromator, where it is scattered at various angles into different wavelengths.
Because of the limited spectral bandwidth, which can be as low as 1–2 nm, the amount of light reaching the detector of a spectrophotometer is typically much lower than that available for a colorimeter. As a consequence, a more sensitive detector is required. The photoelectric detector’s electrical signal is amplified electronically and shown in digital form. These instruments have a significant benefit in terms of measuring speed. There are two types of optical configurations for spectrophotometers:
Singlebeam spectrophotometers:
A singlebeam spectrophotometer with a fixedwavelength monochromator is the most basic spectrophotometer. The wavelength range is 340–625nm (950nm when using a redsensitive detector) with a fixed effective bandwidth of 20nm, while better versions with bandwidths of 2–8nm are also accessible.
A light source, a collimator, a monochromator, a wavelength selector, a cuvette for sample solution, a photoelectric detector, and a digital monitor or meter are all part of it. The monochromator is a 600-lines-per-mm diffraction grating. The spectral bandwidth is defined by the width of the exit slit and the dispersion of light from the diffraction grating, which in this instrument is 20nm.
To determine the Po value needed for an absorbance calculation, the instrument is calibrated with a reference cell containing only solvent. The instruments are battery powered and compact, making them easy to bring into the field. Since manually scanning the wavelength and recalibrating the spectrophotometer, which can be time consuming, singlebeam spectrophotometers are not ideal for recording spectra.

Doublebeam spectrophotometer:
Through using a doublebeam configuration in the spectrophotometer’s optical path, the drawbacks of fixedwavelength, singlebeam spectrophotometers can be reduced. The light beam is divided into two directions, one of which passes through the sample and the other through a reference norm. A chopper controls the radiation’s course, which alternates between the sample, the blank, and the shutter.
The signal processor resolves the signal approaching the detector into the transmission of the blank, shutter, and sample using the chopper’s known rotation speed. It is possible to change 0 percent T continuously using an opaque shutter. The monochromator’s entrance and exit slits are normally adjusted to monitor the effective bandwidths of these devices, which are generally 0.2–3.0 nm. The scanning monochromator can be used to automatically record spectra. Double-beam instruments are more versatile, useful for both quantitative and qualitative studies, and are less sensitive to light source or detector fluctuations.
Components of Spectrophotometers
A typical spectrophotometer type instrument has the same basic components as that of a colorimeter except that the spectrophotometer has a monochromator in place of filters. The following are the essential components of a typical spectrophotometer:
- A source of radiant energy: Which may be a tungsten lamp, a xenon‐mercury arc, hydrogen or deuterium discharge lamp, LEDs, lasers, etc.
- Monochromator: For the selection of a narrow band of radiant energy. It could be a prism or a diffraction grating or a holographic grating monochromator.
- An optical system: Consisting of lenses, mirrors, slits, diaphragm, etc. for producing a parallel beam of light for passage through a sample contained in an absorption cell (cuvette).
- A detecting system: For the measurement of unabsorbed radiant energy, which could be a barrier‐layer cell, phototube, photomultiplier tube, and photodiodes or photodiode array (PDA).
- A readout system or display: Which may be an indicating meter or a numerical digital display or LCD screen.
References:
- Clinical Chemistry, Immunology and Laboratory Quality Control , Elsevier
- Tietz fundamentals of clinical chemistry and molecular diagnostics, 7th edition
- Compendium of Biomedical Instrumentation, Vol 3
