Table of Contents
Introduction
Semen analysis plays a crucial role in assessing male fertility health. It involves the examination of semen volume, sperm concentration, motility, and morphology. Understanding the composition and characteristics of semen can provide valuable insights into reproductive health.
Composition of semen
Semen is composed of various components:
- Spermatozoa: Approximately 5% of the semen consists of sperm cells, which are essential for fertilization.
- Seminal Fluid: Making up 60% to 70% of the total volume, this fluid provides the medium for sperm transport and nourishment.
- Prostate fluid: Comprising 20% to 30%, this component adds further nourishment and support to the sperm.
- Bulbourethral glands: Contributing about 5% to the mix, these glands play a role in lubrication and semen viability.
Appearance
The visual characteristics of semen can provide valuable insights into reproductive health. Normal semen is typically clear and possesses a faint musty odor. However, increased white turbidity may indicate the presence of white blood cells and potential reproductive tract issues. To ensure accurate analysis, any abnormalities may prompt specimen culture before microscopic inspection. Isolating immature sperm from white blood cells is essential, and a leukocyte esterase strip reagent check can be employed for effective screening. Uncommonly, red blood cells (RBCs) with varying degrees of redness may be observed. The presence of a yellow coloration is attributed to factors such as sperm oxidation, extended abstinence, and medication use.
Liquefaction
The process of liquefaction is vital for accurate semen analysis. Freshly ejaculated sperm tends to coagulate and should ideally liquefy within 30 to 60 minutes. Monitoring the collection time is critical to ensure proper liquefaction, which is essential for accurate testing. If liquefaction has not occurred after 2 hours, enzymes like alpha-chymotrypsin may be used to facilitate the process. Challenges in liquefaction could indicate a deficiency in prostatic enzymes, warranting further investigation.
Volume
Semen volume, typically ranging from 2 to 5 mL, serves as a crucial parameter for assessing male fertility. To determine volume accurately, the material is poured into a calibrated sterile graduated container at 0.1-mL intervals. Prolonged periods of abstinence may lead to reduced volume. Diminished volume is often associated with infertility and might indicate abnormal functioning of semen-producing organs, particularly the seminal vesicles. Additionally, the possibility of inadequate specimen collection should not be overlooked.
Viscosity
Viscosity, referring to the fluid consistency of semen, is closely linked to liquefaction. High viscosity can cause agglomeration of materials, impeding accurate analysis. In a healthy sample, sperm can be easily drawn into a pipette and released in droplets that are neither clumped nor stringy. Proper droplets form a thin chain as they are released. Abnormal viscosity, characterized by long strings exceeding 2 cm, can hinder sperm motility. The Viscosity Test employs a scale ranging from 0 (watery) to 4 (sticky/gel-like), with categories like minimum, moderate, and solid.
pH
The pH level of semen provides insights into the health of the reproductive tract. A normal pH range of 7.2 to 8.0 is considered alkaline. Elevated pH may suggest an underlying infection. pH testing involves adding a semen specimen to a urinalysis reagent strip and comparing the color to a chart. pH test paper can also serve this purpose effectively.
Sperm Concentration/Count
Sperm concentration plays a pivotal role in male fertility. A sperm count of over 20 million per milliliter is considered normal, with values between 10 to 20 million per milliliter falling within borderline ranges. Total ejaculate sperm count is determined by multiplying sperm content with specimen volume. A typical healthy ejaculate should contain over 40 million sperm (around 20 million per milliliter and approximately 2 mL in volume). Sperm concentration is a significant predictor of fertility potential, even when just a single spermatozoon fertilizes an egg.
Sperm Motility
Sperm motility, characterized by forward movement, is essential for successful fertilization. The World Health Organization grades sperm motility based on categories A, B, C, and D, reflecting different degrees of mobility. To be considered optimal, 50% or more of sperm in categories A, B, and C should display mobility within an hour, with 25% or more exhibiting progressive motility. Challenges such as immobile sperm or aggregations necessitate further tests to assess sperm viability.
Sperm Motility Grading
| Grade | WHO Criteria | |
| 4 | A | Rapid, Straight-line motility |
| 3 | B | Slower speed, some lateral movement |
| 2 | B | Slow forward progression, noticeable lateral movement |
| 1 | C | No forward progression |
| 0 | D | No Movement |
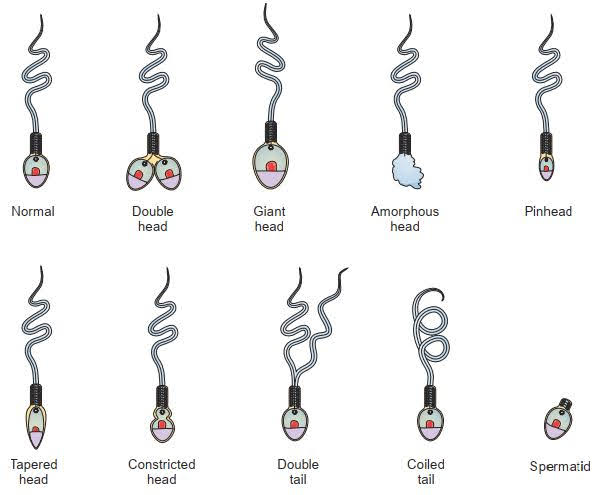
Sperm Morphology
The shape and structure of sperm play a significant role in fertilization success. Evaluation of head, collar, midpiece, and tail forms provides insights into sperm morphology. A normal sperm possesses an oval head measuring approximately 5 μm in length and 3 μm in width. Defects in head morphology can hinder ovum penetration, while abnormalities in other segments like the midpiece and tail impact motility.
The normal sperm has an oval-shaped head that measures approximately 5 μm length by 3 μm width and a flagellar neck that measures around 45μm in thickness. The acrosomal cap, which is positioned at the tip of the head and contains enzymes, is essential for ovum penetration. The acrosomal shield will protect approximately half of the head and two-thirds of the sperm nuclei. The neckpiece is made up of the head, which is joined to the tail, and the midpiece. The midpiece is the thickest part of the tail because it is surrounded by a mitochondrial sheath that allows the tail to move.
Normal sperm morphology levels vary depending on the technique of measurement, ranging from standard forms more than 30% when using normal criteria to standard forms larger than 14% while using strict standards.

Other Examinations of Semen
Fructose
Seminal fructose levels indicate androgen sufficiency, with decreased levels associated with insufficiency. Low fructose production could result from inadequate testosterone or seminal vesicle issues. The resorcinol technique helps assess fructose levels accurately.
Antibodies to Spermatozoa
Spermatozoal antibodies can impact fertility and are assessed through laboratory tests. The role of these antibodies in infertility is established, but more research is needed.
References:
- Concise Book of Medical Laboratory Technology Methods and Interpretations by Ramnik Sood
- Urinalysis and Body Fluids 5th Ed by Strasinger SK and Di Lorenzo MS
