Table of Contents
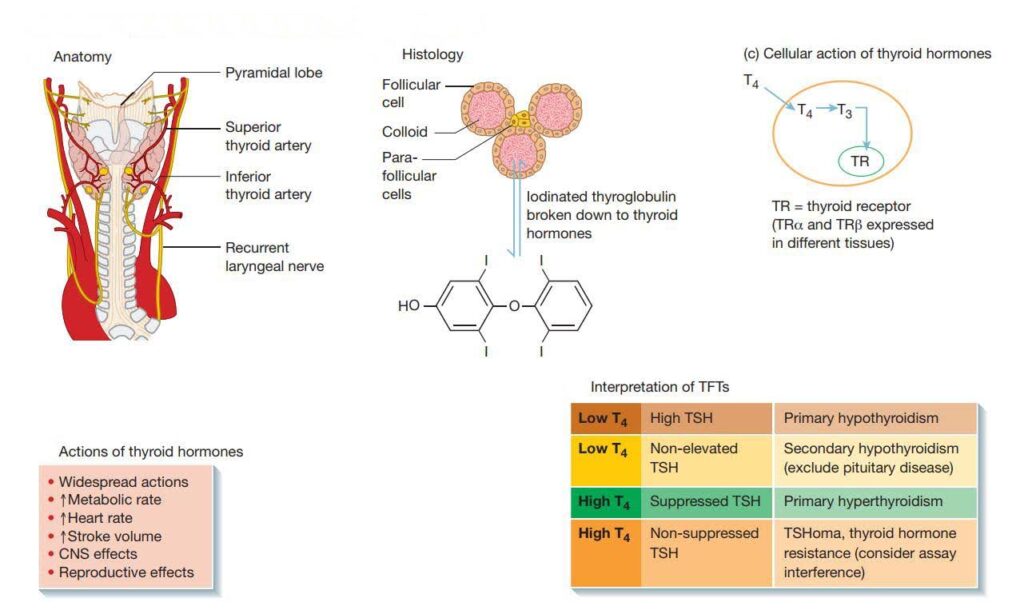
The thyroid gland plays a crucial role in our body’s metabolic activities. It consists of colloid, which contains iodinated thyroglobulin, a large molecule synthesized by follicular cells. Thyroxine, also known as T4, is produced from thyroglobulin and processed in colloid. Additionally, neuroendocrine cells called parafollicular or C cells release calcitonin, which serves as a biomarker for diagnosing medullary thyroid carcinoma. Thyroid disorders can be classified into hypothyroidism (underactive thyroid) and hyperthyroidism (overactive thyroid), leading to symptoms like weight gain, lethargy, and heart rate fluctuations.
To better understand thyroid function, it’s important to know that T4 and triiodothyronine (T3) are produced from the amino acid tyrosine. T4 is the primary hormone circulating in our body, and it gets converted into T3, which is more potent and has a shorter half-life. Thyroxine binding globulin (TBG), transthyretin, and albumin in our bloodstream bind strongly to thyroid hormones. It’s crucial to note that thyroid receptors (TR) only bind to the free hormone. These receptors are divided into two types: TRα and TRβ, which are expressed differently in various organs. Rarely, mutations in TR can lead to thyroid hormone resistance. Local activities of thyroid hormones on tissues are regulated by de-iodinase enzymes (DIO 1, 2, and 3), which activate and deactivate the hormones.
How to Interpret Thyroid Function Test Results
To interpret thyroid function test results accurately, understanding the feedback axis is key. The thyroid gland operates through a classic negative feedback system. Thyrotropin Releasing Hormone (TRH) stimulates the pituitary gland to release thyroid-stimulating hormone (TSH), which, in turn, triggers the secretion of T3 and T4. The peripheral effects of T3 and T4 are governed by TRα and TRβ. Since thyroid hormones have little circadian rhythmicity and are not pulsatile, basal levels are sufficient for interpretation, and dynamic tests are typically unnecessary.
What is Hypothyroidism? Causes and Symptoms
Thyroid disorder, which is also autoimmune in nature, causes primary hypothyroidism. Reduced circulating T3 and T4 levels, as well as a compensatory increase in TSH, characterize this condition. Secondary hypothyroidism is distinguished by low T3/T4 levels and non-elevated TSH – sometimes TSH is normal rather than low – which is triggered by TSH deficiency, which is normally caused by pituitary disease.
Primary hypothyroidism
The most common causes are thyroid antibodies, lymphocytic infiltration, fibrosis, and atrophy, as well as gland enlargement with goitre (Hashimoto’s thyroiditis). Pregnancy may cause acute or chronic hypothyroidism after birth, which may be misdiagnosed as postpartum depression (post-partum thyroiditis). In developing nations, iodine deficiency is a preventable cause of neonatal hypothyroidism, which causes severe mental retardation (cretinism). A unusual genetic mutation in thyroid hormone synthesis can trigger hypothyroidism in children (familial thyroid dyshormonogenesis). Hypothyroidism is caused by two drugs: amiodarone and lithium. Iatrogenic hypothyroidism is caused by the intentional treatment of thyroid disease (e.g. surgery, RAI) or the inadvertent disturbance of thyroid activity caused by radiation to the head and neck region.
Secondary hypothyroidism
TSH dysfunction caused by hypothalamic–pituitary disease causes secondary hypothyroidism, which is far less frequent than primary hypothyroidism. Low fT4 and non-elevated TSH are signs of secondary hypothyroidism, which may warrant a thorough examination of the pituitary gland.
Subclinical hypothyroidism
A typical fT4 with an elevated TSH is referred to as subclinical hypothyroidism. If patients are asymptomatic, therapy may not be necessary; 10–15 percent of patients’ thyroid function returns to normal after repeat monitoring. Because of the high risk of progression to frank hypothyroidism, guidelines prescribe beginning thyroxine even though patients are asymptomatic if TSH is >10 mIU/L. Treatment can also be considered in women considering birth, on a trial basis in symptomatic patients, and in patients with severe dyslipidaemia at lower levels of TSH elevation (TSH 5–10 mIU/L). Patients with healthy thyroid antibodies should have a TFT every year and make sure they don’t develop excessive hypothyroidism.
Hyperthyroidism: Symptoms, Causes, and Treatments
Increased circulating T3 and T4, as well as suppressed TSH due to negative feedback, characterize primary hyperthyroidism. Rare circumstances or assay intervention should be addressed if TSH is not inhibited in the sense of hyperthyroidism. In this case, the clinical picture is critical in determining whether Subclinical hypothyroidism exists.
The sympathetic nervous system is overactive in hyperthyroidism, resulting in a variety of symptoms. Weight loss (often accompanied with a rise of appetite), insomnia and irritability, nausea, heat aversion, palpitations, and resting tremor are all common symptoms. Pruritus, elevated bowel frequency and loose movements, menstrual discomfort, and decreased fertility are all typical hyperthyroidism symptoms.
Resting tachycardia (sinus rhythm or atrial fibrillation), warm peripheries, resting tremor, hyper-reflexia, and lid lag are all common symptoms of hyperthyroidism. Because of increased sympathetic tone in the upper eyelid, lid lag may occur in any cause of hyperthyroidism. Graves’ condition is characterized by lid retraction and proptosis. Hypertension and a flow murmur are possible symptoms of a hyperdynamic circulation. Patients with hyperthyroidism frequently appear anxious and hyperkinetic (the so-called “thyroid affect”).
Graves’ disease
Thyroid eye disorder is one of the specific clinical symptoms of Graves’ disease. Skin alterations (dermopathy) characterized by pre-tibial myxoedema, as well as nail changes similar to clubbing, are more uncommon extra-thyroidal symptoms (thyroid acropachy). Cross-reactivity with TSH receptors in the back of the orbit and on the skin causes these symptoms.
Goitre
Thyroid enlargement is referred to as goitre. Graves’ disease goitres are usually flat, symmetrical, and vascular, and palpation and auscultation also reveal a rush and bruit. The vascularity of nodular goitres is lower, and dominant nodules can be clinically palpable. There may be a single nodule or several nodules.
Thyroid disease and the heart
Hyperthyroidism may manifest as a heart attack or stroke. Supraventricular tachycardia (SVT) or rapid atrial fibrillation is the most frequent acute presentation (AF). Thyrotoxic cardiomyopathy, which is more prevalent in Graves’ disease, is seen in a smaller percentage of patients. Thyroid storm is an uncommon medical emergency characterized by high-output heart dysfunction and agitation. It has a high mortality rate which necessitates a lot of treatment.
Laboratory Diagnosis of Thyroid disorders
T3, T4 and TSH
Hyperthyroidism is characterized by elevated free T4 (fT4) and free T3 (fT3) levels, as well as undetectable TSH levels. T3 toxicosis is described as an increase in fT3 without a decrease in TSH. Subclinical hyperthyroidism is described by a normal fT4/fT3 ratio and a suppressed TSH, indicating autonomous thyroid function. The occurrence of elevated fT4 and fT3 in the absence of TSH suppression is rare and needs further study.
Thyroid antibodies
Thyroid antibodies may validate Graves’ disease, which could be clinically noticeable on inspection. Thyroid peroxidase antibodies (TPO) are autoimmune thyroid disorder non-specific markers. TSH antibody activating antibodies are more specific and can be useful in specific health conditions, such as breastfeeding, as well as confirming a Graves’ disease diagnosis.
Imaging
Thyroid ultrasound (US) will help confirm nodular thyroid disorder, but it can’t tell you how active the thyroid gland is. Nuclear imaging (technetium or iodine uptake isotope scan) aids in the diagnosis of hyperthyroidism by determining the role of the thyroid gland and thus the cause of hyperthyroidism. In Graves’ disease, uptake increases uniformly, but in nodular disease, uptake increases only in the autonomous nodule (s).
Factors affecting thyroid results
There are several factors that can affect the results of thyroid function tests (TFTs), leading to variations in thyroid hormone levels. It’s important to consider these factors when interpreting the test results. Here are some key factors that can influence TFTs:
- Non-Thyroidal Disease (Sick Euthyroid Syndrome): During acute illnesses or hospitalizations, TFTs can be impacted, causing changes in thyroid hormone levels. This condition, known as sick euthyroid syndrome, often results in central TSH suppression, but various patterns of outcomes can be observed.
- Medications: Certain medications can interfere with thyroid function and alter TFT results. For example, lithium, a medication used for bipolar disorder, can inhibit the release of thyroid hormones, leading to hypothyroidism. Amiodarone, a medication for heart rhythm disorders, can cause both hypo- and hyperthyroidism due to its high iodine content.
- Pregnancy: Thyroid function undergoes changes during pregnancy, especially in the first trimester. The hormone human chorionic gonadotropin (hCG) produced during pregnancy can mildly stimulate the thyroid, leading to a decrease in TSH levels. However, thyroid hormone levels generally remain within the normal range in most pregnant women.
- Age: Thyroid function may vary with age. Older individuals may have slightly higher TSH levels and lower free T4 levels compared to younger individuals, even within the normal range.
- Fasting and Time of Day: TFTs are usually conducted in the morning after an overnight fast. Fasting or the time of day when the test is performed can influence TSH levels.
- Iodine Intake: Adequate iodine intake is essential for proper thyroid function. Both excessive and insufficient iodine intake can affect thyroid function. In regions with iodine deficiency, the risk of hypothyroidism and goiter is increased.
- Autoimmune Conditions: Autoimmune disorders, such as Hashimoto’s thyroiditis and Graves’ disease, can cause fluctuations in thyroid hormone levels.
- Certain Medical Conditions: Certain medical conditions, including chronic kidney disease, liver disease, and pituitary disorders, can impact thyroid function.
- Lab Techniques and Assay Interference: Variations in laboratory techniques and potential assay interference can contribute to discrepancies in TFT results.
To ensure accurate interpretation, it is important to consider these factors and evaluate TFTs in the context of the patient’s overall health and medical history. Performing thyroid function testing in an outpatient setting, when the patient is in good health and not during acute illness or hospitalization, can help minimize the influence of non-thyroidal factors and provide a more reliable evaluation of thyroid function.
References:
- Vargatu I. (2016). WILLIAMS TEXTBOOK OF ENDOCRINOLOGY. Acta Endocrinologica (Bucharest), 12(1), 113
- Wartofsky L, Dickey RA. The evidence for a narrower thyrotropin reference range is compelling. J Clin Endocrinol Metab. 2005;90(9):5483-5488.
- Burch HB, Cooper DS. Management of Graves disease: A review. JAMA. 2015;314(23):2544-2554.
- Glinoer D. The regulation of thyroid function during normal pregnancy: Importance of the iodine nutrition status. Best Pract Res Clin Endocrinol Metab. 2004;18(2):133-152.
- Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: Implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92(12):
