The Nagler reaction, also known as the Nagler test, is a classical yet clinically significant phenotypic laboratory test used for the identification and confirmation of Clostridium perfringens based on its ability to produce lecithinase C (alpha toxin). The test relies on the neutralization of lecithinase activity by a specific antitoxin, allowing reliable differentiation of C. perfringens from other lecithinase-producing anaerobic bacteria.
Clostridium perfringens is a Gram-positive, spore-forming, obligate anaerobic bacillus responsible for serious conditions such as gas gangrene (clostridial myonecrosis), necrotizing soft-tissue infections, food poisoning, and enteritis necroticans. Because these infections can progress rapidly and are associated with high morbidity and mortality, prompt laboratory diagnosis is essential to guide early surgical and antimicrobial management.
Although modern molecular and mass-spectrometry techniques have enhanced bacterial identification, the Nagler reaction remains clinically relevant, particularly in routine diagnostic laboratories, resource-limited settings, and as a confirmatory test demonstrating toxin activity, which directly correlates with pathogenic potential.
Laboratory Diagnosis of Clostridium perfringens
Clinical Diagnostic Relevance
Accurate identification of C. perfringens is crucial in patients presenting with:
- Rapidly spreading wound infections
- Muscle necrosis with gas formation
- Severe abdominal symptoms following food ingestion
- Hemolysis-associated septicemia
The organism’s pathogenicity is primarily mediated by alpha toxin (lecithinase C), making toxin-related diagnostic tests particularly valuable.
Routine Diagnostic Approach
Laboratory diagnosis typically involves a combination of:
- Direct microscopy: Large, box-shaped Gram-positive bacilli with blunt ends
- Anaerobic culture on enriched media
- Hemolysis pattern: Characteristic double-zone hemolysis on blood agar
- Biochemical reactions
- Toxin detection and confirmation, including the Nagler reaction
Among these, the Nagler test provides phenotypic confirmation of alpha-toxin production, strengthening diagnostic confidence.
Principle of the Nagler Reaction
The Nagler reaction is based on two interrelated mechanisms:
- Hydrolysis of lecithin by lecithinase C (phospholipase C)
- Neutralization of lecithinase activity by a specific antitoxic serum
Egg yolk agar contains lecithin, a phospholipid substrate. When lecithinase C is produced by C. perfringens:
- Lecithin is broken down into insoluble diglycerides
- These precipitate in the agar
- A visible opaque or opalescent halo forms around bacterial growth
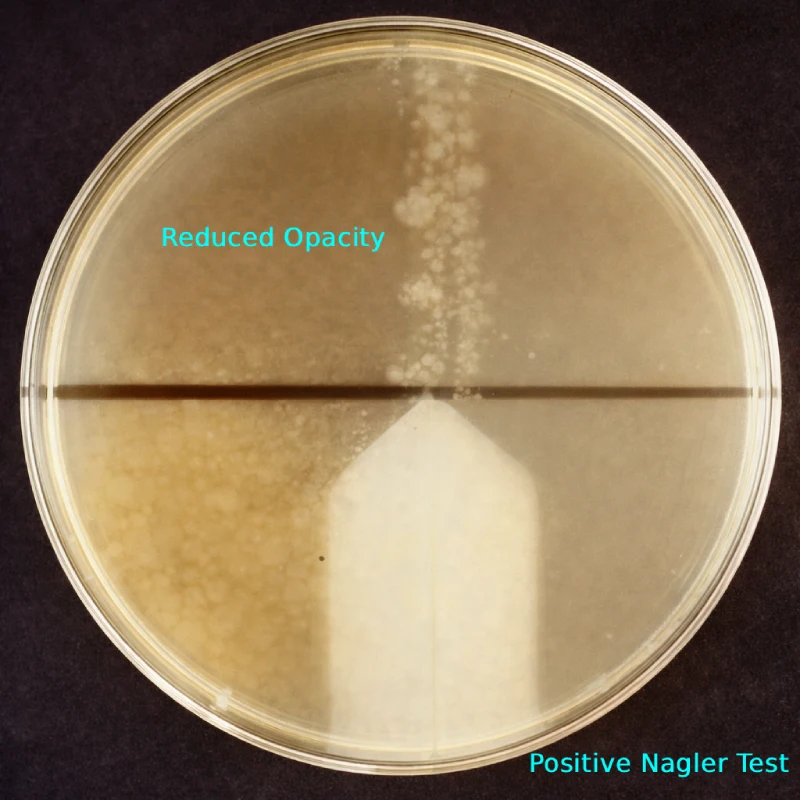
When specific C. perfringens type A antitoxin is applied, it inactivates lecithinase C, leading to disappearance or marked reduction of opacity. This antitoxin-mediated inhibition is the defining feature of the Nagler reaction.
Lecithinase Test vs Nagler Reaction
Lecithinase C (Phospholipase C) Activity
The lecithinase test detects the ability of an organism to hydrolyze lecithin present in egg yolk–containing media.
- Lecithin hydrolysis produces insoluble diglycerides
- These form a diffuse opacity in the surrounding medium
Several organisms produce lecithinase C, including:
- Clostridium perfringens
- Clostridium oedematiens (formerly C. novyi)
- Clostridium sordellii
- Clostridium bifermentans
- Clostridium absonum
Because multiple species can yield a positive lecithinase reaction, lecithinase activity alone is not species-specific and serves only as a screening indicator of potential toxigenicity.
Nagler Reaction: Antitoxin-Based Confirmation
The Nagler reaction is a specific inhibition test that confirms whether lecithinase activity is due to C. perfringens alpha toxin.
- Lecithinase C produces opacity on egg yolk agar
- Specific antitoxin neutralizes alpha toxin
- Opacity is inhibited only in the antitoxin-treated area
This antigen–antibody neutralization differentiates C. perfringens from other lecithinase-producing organisms and provides confirmatory diagnostic evidence.
Key Differences Between Lecithinase Test and Nagler Reaction
| Feature | Lecithinase Test | Nagler Reaction |
| Detects lecithin hydrolysis | Yes | Yes |
| Uses specific antitoxin | No | Yes |
| Species-specific for C. perfringens | No | Yes |
| Diagnostic role | Screening | Confirmatory |
| Clinical relevance | Limited | High |
Media Used for the Nagler Reaction
Egg Yolk Agar
Egg yolk agar is the standard medium for the Nagler test and provides:
- Lecithin as substrate
- Nutritional support for anaerobes
- Clear visualization of enzyme activity
Alternative Media
- Serum agar
- Lactose–egg-yolk–milk agar, which additionally:
- Detects lactose fermentation
- Aids differentiation from other Nagler-positive clostridia
Instruments and Equipment
Essential Laboratory Instruments
- Anaerobic jar or anaerobic chamber
- Incubator (35–37 °C)
- Egg yolk agar plates
- C. perfringens type A antitoxin
- Sterile bacteriological loop or straight wire
- Hockey stick spreader or micropipette
Advantages
- Simple and cost-effective
- Suitable for routine laboratories
- No advanced molecular infrastructure required
Limitations
- Requires strict anaerobic conditions
- Visual interpretation may be subjective
- Antitoxin availability may be limited
Procedure of the Nagler Reaction
Plate Preparation
- Prepare an egg yolk agar plate
- Apply 60 µL of C. perfringens antitoxin to one half
- Spread evenly and allow to dry
- Clearly mark the antitoxin-treated side
Inoculation
- Streak the test organism in a straight line
- Start from the antitoxin-free side and extend across the antitoxin side
- Inoculate control organisms similarly
Incubation
- Incubate anaerobically
- Temperature: 35–37 °C
- Duration: 24–48 hours
Quality Control
Recommended Control Strains
- Positive control: Clostridium perfringens (e.g., NCTC 8359)
- Negative control: Clostridium difficile (e.g., NCTC 11204)
Quality control ensures accuracy of reagents, media, and interpretation.
Interpretation of Results
Positive Nagler Reaction
- Opalescent halo on antitoxin-free side
- Marked reduction or absence of opacity on antitoxin side
- Indicates lecithinase C neutralization
- Confirms C. perfringens
Negative Result
- Opacity present on both sides
- Lecithinase activity not neutralized
- Suggests other clostridial species
Interpretation Challenges
- Enzyme diffusion through agar
- Heavy inoculum causing partial inhibition
- Comparison with uninoculated controls is essential

Clinical Significance
- Confirms alpha-toxin–producing C. perfringens
- Supports diagnosis of gas gangrene
- Assists in food poisoning outbreak investigations
- Influences urgent clinical and surgical decisions
Limitations of the Nagler Test
- Partial cross-reactions may occur
- Cannot quantify toxin levels
- Does not replace molecular toxin typing
- Subjective visual interpretation
Precautions and Best Practices
- Use freshly prepared egg yolk agar
- Avoid heavy inoculation
- Maintain strict anaerobic conditions
- Always include control strains
- Clearly label antitoxin-treated areas
Conclusion
The Nagler reaction remains a robust, reliable, and clinically valuable diagnostic test in anaerobic bacteriology. By demonstrating lecithinase C activity and its neutralization by specific antitoxin, it provides confirmatory identification of Clostridium perfringens. While advanced molecular methods offer enhanced sensitivity, the Nagler test continues to serve as a cost-effective, educational, and practical tool that supports diagnosis and timely patient management.
References
- Murray, P. R., Rosenthal, K. S., & Pfaller, M. A. (2023). Medical Microbiology (10th ed.). Elsevier.
- Koneman, E. W., et al. (2022). Color Atlas and Textbook of Diagnostic Microbiology. Lippincott Williams & Wilkins.
- Public Health England. (2014). UK Standards for Microbiology Investigations: Nagler Test.
- Centers for Disease Control and Prevention. (2024). Clostridial Myonecrosis (Gas Gangrene).
- Nagler, F. P. O. (1939). Observations on a reaction between the lethal toxin of Clostridium welchii and human serum. British Journal of Experimental Pathology.
