Table of Contents
Introduction
Leishmaniasis, a vector-borne disease caused by protozoan parasites of the genus Leishmania, is transmitted through bites of infected female sandflies. As a neglected tropical disease, it affects millions globally, necessitating accurate laboratory diagnosis for effective management. The World Health Organization (WHO) estimates 12 million people are affected, with approximately 1.5 million new cases of cutaneous leishmaniasis (CL) and 500,000 cases of visceral leishmaniasis (VL) annually (World Health Organization, 2023; Garcia, 2016). Untreated VL is nearly always fatal, while CL and mucocutaneous leishmaniasis (ML) cause significant morbidity, including disfigurement and social stigma.
Laboratory diagnosis is critical for confirming infection, guiding treatment, assessing disease severity, monitoring therapeutic response, and supporting epidemiological surveillance. Accurate diagnosis is particularly vital in endemic regions and among immunocompromised populations, such as those with HIV, where atypical presentations complicate clinical assessment (Centers for Disease Control and Prevention, 2024). This article provides a comprehensive guide to laboratory methods for diagnosing leishmaniasis, detailing principles, procedures, clinical significance, limitations, and precautions. It integrates traditional techniques like microscopy and culture with advanced molecular tools, aligning with guidelines from WHO, the Infectious Diseases Society of America (IDSA), and the Centers for Disease Control and Prevention (CDC).
Overview of Leishmaniasis
Epidemiology

Leishmaniasis is endemic in 88 countries across tropical and subtropical regions, including South Asia, East Africa, South America, and the Mediterranean. Over 20 Leishmania species are pathogenic to humans, with L. donovani, L. infantum, L. major, L. tropica, and L. braziliensis being the most clinically significant (Garcia, 2016). Transmission occurs primarily via female sandflies—Phlebotomus species in the Old World (Africa, Asia, Europe) and Lutzomyia in the New World (Americas). Zoonotic reservoirs, including dogs, rodents, and foxes, sustain transmission cycles, while anthroponotic VL is notable in India and Sudan (Alvar et al., 2012).
Recent outbreaks, such as in Sudan (2021–2022) and Spain (2009–2012), highlight the disease’s resurgence due to climate change, urbanization, and conflict-driven migration (Arce et al., 2022). Immunocompromised individuals, particularly those with HIV, face higher risks of severe or disseminated disease, necessitating robust diagnostic strategies (Centers for Disease Control and Prevention, 2024). Laboratory methods must account for geographic variations, as species influence clinical presentation and diagnostic yield.
Life Cycle, Vector, and Host
Leishmania has a digenetic life cycle involving two main forms: the promastigote, which is flagellated and develops in the midgut of the female sandfly vector (Phlebotomus or Lutzomyia), and the amastigote, the non-motile intracellular form found in the mononuclear phagocytes of the mammalian host. Infection begins when an infected sandfly injects promastigotes during a blood meal. These are phagocytosed by macrophages and transform into amastigotes, which multiply and infect other cells. When another sandfly feeds, it ingests these infected macrophages, completing the cycle. The site of infection and the clinical form depend on the Leishmania species and the host’s immune response, which directly influences specimen selection and diagnostic approach.

Clinical Forms
Leishmaniasis presents in three primary forms, each requiring tailored diagnostic approaches:
- Cutaneous Leishmaniasis (CL): Characterized by self-healing ulcers or nodules on exposed skin, caused by species like L. major (Old World) and L. mexicana (New World). Lesions vary from papules to crusted ulcers, often resolving within months but leaving scars.
- Mucocutaneous Leishmaniasis (ML): Caused by L. braziliensis complex, ML leads to destructive lesions in mucosal tissues (e.g., nose, mouth) years after initial CL infection. It is prevalent in Latin America and requires early diagnosis to prevent disfigurement.
- Visceral Leishmaniasis (VL, or Kala-azar): A systemic, life-threatening form caused by L. donovani or L. infantum, affecting the liver, spleen, and bone marrow. Symptoms include prolonged fever, hepatosplenomegaly, weight loss, and pancytopenia, with near-100% mortality if untreated.
Post-kala-azar dermal leishmaniasis (PKDL), a sequela of VL, manifests as hypopigmented macules or nodules, particularly in India and East Africa. Atypical presentations in immunocompromised patients, such as diffuse cutaneous or visceral dissemination, underscore the need for laboratory confirmation (Bern et al., 2016).
Diagnosing Human Infections
The core principle of diagnosing leishmaniasis is detecting Leishmania parasites (amastigotes in human tissues, promastigotes in vectors or culture) or host immune responses (antibodies, antigens, or DNA). Diagnostic methods are selected based on clinical form, parasite burden, disease stage, host immunity, and resource availability (Pace, 2022). For CL and ML, direct visualization of amastigotes in skin lesions is often sufficient due to high parasite loads. In VL, where parasites are sparse in peripheral tissues, serological and molecular tests are prioritized.
Diagnosis integrates clinical history, epidemiological context, and laboratory findings. For example, VL patients often present with hypergammaglobulinemia, prompting serological screening, while CL diagnosis relies on lesion sampling. Methods must balance sensitivity, specificity, and feasibility, especially in resource-limited settings, as emphasized by WHO guidelines (World Health Organization, 2023).
Structure of Leishmania Parasite
Leishmania exists mainly in two morphological forms relevant to diagnosis: the amastigote, a small (2–5 μm), oval, non-flagellated form found inside host macrophages, characterized by a nucleus and a rod-shaped kinetoplast; and the promastigote, a larger (10–20 μm), elongated, flagellated form present in the sandfly vector and culture media. Recognizing these forms microscopically is fundamental for accurate diagnosis and differentiation from other intracellular pathogens.

Specimen Collection and Handling
For Cutaneous and Mucocutaneous Leishmaniasis
Obtain samples from active lesion margins, where amastigotes concentrate within macrophages, avoiding necrotic centers that yield debris and contaminants.
Procedure:
- Clean the lesion with 70% alcohol to reduce bacterial contamination.
- Collect samples using:
- Dermal Scraping: Scrape lesion edges with a sterile scalpel blade.
- Fine-Needle Aspiration (FNA): Aspirate with saline using a 25-gauge needle.
- Punch Biopsy: Obtain a 3–4 mm biopsy from the lesion border under local anesthesia.
- Prepare multiple smears on slides, fix with methanol for microscopy, and divide biopsy portions for culture and histopathology (Garcia, 2016).
- Transport smears fixed; store culture samples in sterile saline or media at room temperature.

Direct sampling confirms Leishmania infection, differentiates from mimics like sporotrichosis or basal cell carcinoma, and enables species identification via molecular tools, guiding treatment.
Low parasite density in chronic or healing lesions reduces yield. Secondary bacterial or fungal infections may contaminate cultures.
Use aseptic techniques to prevent contamination. Wear personal protective equipment (PPE) to avoid needlestick injuries. Handle specimens as biosafety level 2 (BSL-2) materials. Label with biohazard warnings and comply with transport regulations.
For Visceral Leishmaniasis
Target reticuloendothelial tissues (bone marrow, spleen, lymph nodes) where amastigotes proliferate, as peripheral blood has lower sensitivity.
Procedure:
- Bone Marrow Aspiration: Preferred method (80–85% sensitivity). Aspirate 1–2 ml from the iliac crest or sternum under local anesthesia. Prepare smears and inoculate culture media.
- Splenic Aspiration: Highly sensitive (95–98%) but invasive; perform under ultrasound guidance to minimize hemorrhage risk.
- Peripheral Blood (Buffy Coat): Non-invasive but less sensitive (50–65%, higher at night due to parasite periodicity). Centrifuge blood to isolate buffy coat for smears or PCR (Garcia, 2016; Arce et al., 2022).
- Transport smears fixed in methanol; store culture or PCR samples at 4–8°C if processing is delayed.
Confirms systemic infection, essential for immunocompromised patients (e.g., HIV co-infection) where atypical dissemination occurs.
Invasive procedures carry risks (e.g., bleeding, infection). False negatives occur in early disease or low parasitemia.
Precautions: Obtain informed consent for invasive procedures. Monitor for post-aspiration bleeding. Use sterile equipment and handle as BSL-2. Avoid freezing smears to preserve morphology.
General Handling and Transport
- Preserve parasite viability and morphology for accurate diagnosis.
- Fix smears with methanol immediately. Transport culture samples in Novy-MacNeal-Nicolle (NNN) or Schneider’s medium at room temperature. For PCR, store samples in ethanol or frozen at -20°C.
- Proper handling ensures reliable results, reducing misdiagnosis in endemic settings.
- Delays degrade sample quality, especially in remote areas with limited cold-chain infrastructure.
- Label specimens with patient details and biohazard warnings. Comply with international shipping regulations for infectious substances (Centers for Disease Control and Prevention, 2024).
Microscopic Examination
Principle
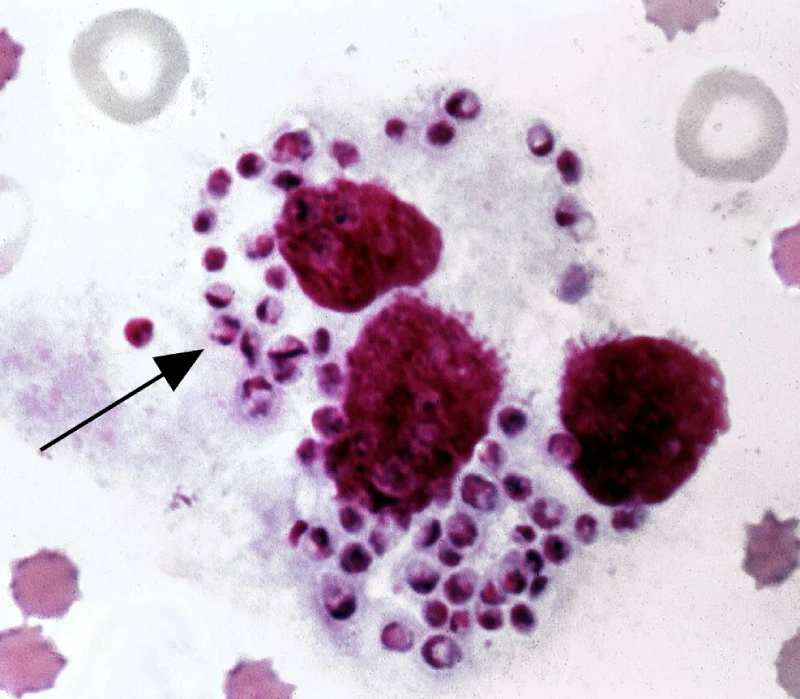
Microscopy detects intracellular amastigotes (2–5 μm, ovoid, with a nucleus and rod-shaped kinetoplast) in stained smears, distinguishing Leishmania from mimics like Histoplasma or Toxoplasma.
Procedure
- Air-dry smears and fix with methanol for 1–2 minutes.
- Stain with Giemsa (30–60 minutes, diluted 1:10) or Wright’s stain.
- Examine under oil immersion (1000x magnification) for amastigotes within macrophages or free in tissue.
- For biopsies, use hematoxylin-eosin (H&E) staining for histopathological sections (Garcia, 2016; Pace, 2022).
Clinical Significance
- Gold standard for CL and ML due to high parasite loads (sensitivity >90%).
- Rapid (results within hours) and cost-effective, ideal for resource-limited settings.
- In VL, confirms heavy parasite burdens in bone marrow or splenic aspirates, aiding prognosis.
Limitations
- Operator-dependent; requires trained microscopists to identify amastigotes accurately.
- Low sensitivity in VL (50–80% for bone marrow, lower for blood).
- False negatives in chronic or low-burden infections; kinetoplast may be obscured in tissue sections.
Precautions
- Scan at least 1,000 fields to rule out infection.
- Use positive controls to validate staining and microscopy.
- Fix slides before handling to reduce biohazard risk (BSL-2).
- Avoid over-staining, which obscures parasite morphology.
Culture Methods
Principle
Culturing transforms amastigotes into motile promastigotes in nutrient-rich media, increasing diagnostic sensitivity and enabling species identification.
Procedure
- Inoculate specimens (lesion aspirates, bone marrow, or biopsies) into NNN or Schneider’s insect medium supplemented with 30% fetal bovine serum.
- Incubate at 22–26°C to mimic sandfly conditions.
- Examine weekly for 4 weeks via wet mount or Giemsa-stained smears for motile promastigotes (15–20 μm, flagellated) (Garcia, 2016).
- Subculture positive isolates for downstream molecular or isoenzyme analysis.
Clinical Significance
- Enhances sensitivity (up to 90% when combined with microscopy), especially in chronic CL or VL with inconclusive smears.
- Provides viable parasites for speciation, critical for treatment decisions (e.g., antimonial resistance in L. tropica).
- Useful in research settings for studying parasite biology.
Limitations
- Time-consuming (1–4 weeks for results), limiting clinical utility in acute cases.
- Prone to bacterial or fungal contamination, especially from skin lesions.
- Requires specialized media, incubators, and trained personnel, often unavailable in endemic areas.
Precautions
- Collect samples aseptically and transport promptly in appropriate media.
- Add antibiotics (e.g., penicillin, streptomycin) to culture media if contamination is suspected.
- Maintain quality control with known Leishmania strains.
- Handle as BSL-2 due to infectious promastigotes.
Serological Tests
Principle
Serological tests detect antibodies against Leishmania antigens, particularly useful in VL where direct parasite detection is challenging due to low peripheral parasitemia.
Procedure
- Collect serum from venous blood.
- Perform tests:
- Enzyme-Linked Immunosorbent Assay (ELISA): Detects IgG against crude or recombinant antigens.
- Indirect Fluorescent Antibody (IFA): Measures antibody binding to fixed promastigotes.
- Direct Agglutination Test (DAT): Mix serum dilutions with freeze-dried antigen; read agglutination after 18 hours.
- rK39 Dipstick Test: Rapid immunochromatographic test detecting anti-rK39 IgG in 10–20 minutes (Boelaert et al., 2019).
- Interpret results based on manufacturer cutoffs or titers.
Clinical Significance
- High sensitivity (95–100%) and specificity (>90%) for VL, ideal for screening in endemic areas.
- rK39 dipstick is field-friendly, requiring no equipment, making it valuable for rapid diagnosis during outbreaks.
- Monitors treatment response indirectly via declining antibody titers.
Limitations
- Low sensitivity in CL and ML due to minimal humoral response.
- Cross-reactivity with trypanosomiasis, tuberculosis, or malaria reduces specificity.
- Persistent antibodies post-cure lead to false positives in previously treated patients.
- Poor performance in immunocompromised patients (e.g., HIV co-infection) due to suppressed antibody production.
Precautions
- Confirm positive serology with parasitological or molecular tests.
- Store reagents at 4–8°C and validate kits per manufacturer guidelines.
- Interpret results in the context of clinical and epidemiological data to avoid misdiagnosis.
Molecular Diagnostics
Principle
Polymerase chain reaction (PCR) amplifies Leishmania DNA or RNA, offering high sensitivity and specificity for detecting low parasite loads and identifying species.
Procedure
- Extract DNA from blood, bone marrow, lesion aspirates, or biopsies using commercial kits.
- Perform conventional or real-time PCR targeting conserved regions (e.g., kinetoplast DNA, 18S rRNA, ITS1).
- Use multiplex PCR for species differentiation (e.g., L. donovani vs. L. braziliensis).
- Analyze results via gel electrophoresis (conventional) or fluorescence (real-time) (Pace, 2022).
Clinical Significance
- Sensitivity >95% in VL and CL, even in low-parasitemia or atypical cases.
- Quantifies parasite load, aiding treatment monitoring and relapse detection.
- Species identification informs therapy, especially for drug-resistant strains (e.g., L. tropica).
Limitations
- High cost and need for specialized equipment (thermocyclers, fluorescence detectors).
- Requires trained personnel and clean-room conditions to prevent contamination.
- Lack of global standardization limits adoption in resource-poor settings.
Precautions
- Use negative and positive controls to validate results.
- Perform DNA extraction and amplification in separate areas to avoid cross-contamination.
- Handle specimens as BSL-2 during extraction.
- Validate primers for specificity to Leishmania species.
Other Diagnostic Approaches
Leishmanin Skin Test (Montenegro Test)
Principle: Measures delayed-type hypersensitivity to intradermal injection of killed promastigotes, indicating prior exposure or immunity.
Procedure: Inject 0.1 ml of antigen intradermally; measure induration (>5 mm) at 48–72 hours.
Clinical Significance: Useful for epidemiological surveys to assess population exposure, particularly in CL-endemic areas. Positive in resolved CL or VL, negative in active VL.
Limitations: Not diagnostic for active infection; unavailable in many regions due to lack of standardized antigen.
Precautions: Contraindicated in pregnancy or immunosuppression. Interpret cautiously in co-endemic areas with other protozoal infections.
Antigen Detection
Principle: Detects Leishmania antigens in urine or serum, offering an alternative to antibody-based tests.
Procedure: Use tests like KAtex (latex agglutination for k39 antigen in urine) or ELISA-based antigen assays.
Clinical Significance: Moderate sensitivity (60–80%) for VL; useful in immunocompromised patients where serology fails. Rapid results aid field diagnosis.
Limitations: Less effective for CL or ML; variable performance across populations.
Precautions: Use fresh samples to avoid degradation. Follow kit storage instructions (typically 4–8°C).
Animal Inoculation
Principle: Inoculate clinical specimens into susceptible animals (e.g., hamsters) to amplify Leishmania parasites for detection.
Procedure: Inject lesion aspirates or bone marrow into hamster footpads or peritoneum. Monitor for lesions or organ involvement over weeks to months, followed by microscopy or culture of tissues (Garcia, 2016).
Clinical Significance: Historically used for confirming infection when microscopy or culture fails, particularly in research settings.
Limitations: Slow (weeks to months), ethically controversial, and impractical for routine diagnosis due to animal welfare concerns and lab requirements.
Precautions: Follow ethical guidelines for animal use. Handle as BSL-2 due to infectious material. Rarely used in modern diagnostics.
Differential Diagnosis
Laboratory diagnosis must differentiate leishmaniasis from mimics:
- CL: Mycobacterial infections (e.g., cutaneous tuberculosis), leprosy, sporotrichosis, sarcoidosis, basal cell carcinoma, or fungal infections (e.g., chromomycosis).
- ML: Paracoccidioidomycosis, squamous cell carcinoma, or Wegener’s granulomatosis.
- VL: Malaria, typhoid fever, leukemia, lymphoma, brucellosis, or schistosomiasis.
Combining microscopy, serology, and PCR ensures accurate differentiation, particularly in co-endemic regions or immunocompromised patients (Bern et al., 2016).
Challenges in Diagnosis
Diagnosing leishmaniasis in endemic areas faces multiple hurdles, requiring tailored strategies to overcome them.
Resource Limitations
Many endemic regions, where it lack access to advanced diagnostics like PCR or culture facilities. Microscopy and rK39 dipsticks are often the only available tools, but their sensitivity varies. For example, microscopy requires skilled technicians, and rK39 tests may fail in HIV co-infected patients (Boelaert et al., 2019). Mobile diagnostic units and point-of-care tests are critical for improving access, but funding and infrastructure remain barriers.
Coinfections
HIV-Leishmania co-infection, complicates diagnosis. Immunosuppression reduces antibody production, lowering serology sensitivity, and atypical parasite dissemination (e.g., to skin or lungs) requires invasive sampling or PCR (Alvar et al., 2012). Similarly, co-endemic diseases like malaria or tuberculosis can mimic VL symptoms, necessitating multiple tests for accurate diagnosis.
Drug Resistance
Emerging resistance, particularly to antimonials in L. tropica and L. donovani, underscores the need for species identification. Molecular tests like PCR are essential but often unavailable. Misdiagnosis or delayed speciation can lead to treatment failure, prolonging morbidity (Pace, 2022).
Emerging Diagnostic Tools
Novel technologies like loop-mediated isothermal amplification (LAMP) and CRISPR-based diagnostics offer promise for point-of-care testing. LAMP amplifies DNA without thermocyclers, achieving >90% sensitivity in field trials for VL (Mukhtar et al., 2018). CRISPR-based assays, such as SHERLOCK, detect Leishmania DNA with high specificity but are still experimental (Pace, 2022). Validation and cost reduction are needed for widespread adoption.
Regional Variations
Diagnostic yield varies by region due to species differences. For example, L. infantum in the Mediterranean responds well to bone marrow PCR, while L. braziliensis in South America requires biopsy-based diagnosis for ML. Standardizing protocols across regions remains challenging (Arce et al., 2022).
Conclusion
Laboratory diagnosis of leishmaniasis integrates traditional and modern methods to achieve high accuracy across its diverse clinical forms. Microscopy and culture remain cornerstones for CL and ML, while serology and PCR are critical for VL, particularly in low-resource or complex cases. Each method’s principles, procedures, and limitations must guide its application, with precautions ensuring safety and reliability. Emerging tools like LAMP and CRISPR promise to enhance point-of-care diagnostics, addressing challenges in endemic areas. By combining clinical judgment with robust laboratory testing, clinicians can reduce morbidity, prevent transmission, and support global control efforts for this debilitating disease.
References
- Alvar, J., Vélez, I. D., Bern, C., Herrero, M., Desjeux, P., Cano, J., Jannin, J., & den Boer, M. (2012). Leishmaniasis worldwide and global estimates of its incidence. PLoS One, 7(5), e35671. https://doi.org/10.1371/journal.pone.0035671
- Arce, A., Estirado, A., Ordobas, M., Sevilla, S., Garcia, N., Moratilla, L., de la Fuente, S., Martinez, A. M., Perez, A. M., Aranguez, E., Iriso, A., Sevillano, O., Bernal, J., & Vilas, F. (2022). Re-emergence of leishmaniasis in Spain: Community outbreak in Madrid, Spain, 2009 to 2012. Euro Surveillance, 27(10), 120-130. https://doi.org/10.2807/1560-7917.ES.2022.27.10.2100430
- Bern, C., Maguire, J. H., & Alvar, J. (2016). Complexities of assessing the disease burden attributable to leishmaniasis. PLoS Neglected Tropical Diseases, 10(10), e0005058. https://doi.org/10.1371/journal.pntd.0005058
- Boelaert, M., Verdonck, K., Menten, J., Sunyoto, T., van Griensven, J., Chappuis, F., & Rijal, S. (2019). Rapid tests for the diagnosis of visceral leishmaniasis in patients with suspected disease. Cochrane Database of Systematic Reviews, 6(6), CD009135. https://doi.org/10.1002/14651858.CD009135.pub3
- Centers for Disease Control and Prevention. (2024). Clinical testing and diagnosis for leishmaniasis. https://www.cdc.gov/leishmaniasis/hcp/diagnosis/index.html
- Garcia, L. S. (2016). Diagnostic medical parasitology (6th ed.). ASM Press. https://doi.org/10.1128/9781555819002
- Mukhtar, M., Ali, S. S., Boshara, S. A., Albertini, A., Monedero, A., Bécourt, E., & Gari-Toussaint, M. (2018). Sensitive and less invasive confirmatory diagnosis of visceral leishmaniasis in Sudan using loop-mediated isothermal amplification (LAMP). PLoS Neglected Tropical Diseases, 12(2), e0006264. https://doi.org/10.1371/journal.pntd.0006264
- Pace, D. (2022). Laboratory diagnostics for human Leishmania infections: A polymerase chain reaction gold standard? Parasites & Vectors, 15(1), 399. https://doi.org/10.1186/s13071-022-05524-z
- World Health Organization. (2023). Leishmaniasis fact sheet. https://www.who.int/news-room/fact-sheets/detail/leishmaniasis
