Table of Contents
Separating nucleic acid is an exceedingly essential tool for molecular biology. Until modern technology could be used, nucleic acid separation was a time-consuming and work-intensive process based on multiple extraction and centrifugation steps, often constrained by small yields and low purity of separation products, and not suitable for automation and upscaling. Specifically functionalized magnetic particles were produced over the last few years. Allowing the rapid and efficient purification directly after their extraction from crude cell extracts, together with a suitable buffer system. Centrifugation steps is no longer required.
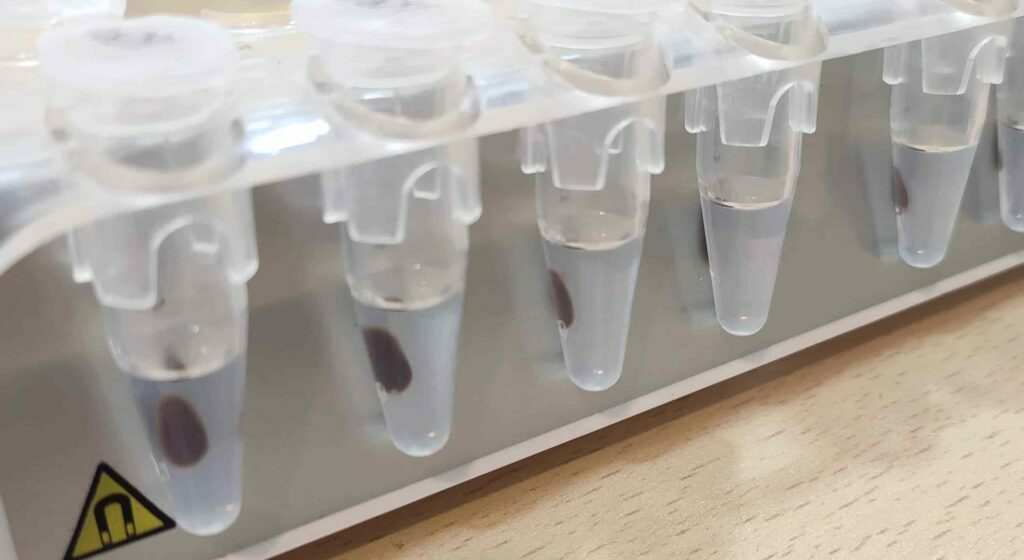
Magnetic separation
Compared with other non-magnetic separation methods, the growing use of magnetic solid carriers in biochemical and molecular biology methods has many advantages. The term ‘magnetic’ means that when put in a magnetic field the aid obtains a magnetic moment. And it may be replaced. In other words, particles which have a magnetic moment can be quickly extracted by applying a magnetic field. It is a fast , easy and effective way to isolate the particles after the nucleic binding or elution step, and a process much less stringent than conventional methods, such as centrifugation, which produces shear forces that can contribute to nucleic acid degradation.
Magnetic carriers with affinity ligands immobilized on them or biopolymers with affinity to the target nucleic acid are used throughout the isolation process. The majority of magnetic carriers are commercially available, although they can also be produced in the lab. Magnetic particles made of various synthetic polymers, biopolymers, porous glass, or magnetic particles based on inorganic magnetic properties like surface-modified iron oxides are examples of these materials. Superparamagnetic particles are particularly well suited because they do not disturb when there is no magnetic field present. These particles will magnetize in a strong magnetic field, but they will lose their magnetism if the field is removed. Magnetic aggregation and clumping of the particles was eliminated during the reaction, ensuring simple particle suspension and uniform nucleic acid isolation.
Magnetic bead DNA extraction
In a nutshell, during the lysis step, Nucleic Acids adhere to the charged surface of a paramagnetic particle in the presence of certain salts. Nucleic acids and other toxins may be gathered with the aid of a magnet. Until the removed nucleic acids in the form of low salt buffer or water are released from the paramagnetic sample, further washes in the presence of alcohol mixed with salt are performed. Without the use of centrifugation or filtration, nucleic acid separation, drying, and elution are achieved.

As the beads adhere to DNA, the underlying magnetic field pulls them to the outside of the surrounding tube, immobilizing them. The bead-bound DNA is retained during the washing steps, while the beads are immobilized. When the elution buffer is used and the magnetic field is removed, the DNA becomes a pure sample that can be quantified and examined.
The ability of Nucleic Acids to reversibly adhere to a charged surface while exposed to such amounts of salts allows them to be distinguished from other undesirable molecules. Magnetic based purification can be used by a variety of surface chemistries and applications. The properties of ligand binding can vary from indiscriminate to sequence- or tag-specific.
| Type | Properties | Applications |
| Carboxylate-modified magnetic beads | Can associate with nucleic acids for direct capture. Surface suitable for conjugation through covalent bonding. Can capture molecules containing amino groups. | Conjugation or direct binding applications: Covalent attachment Affinity purification and pull-down Nucleic acid isolation and purification NGS size selection |
| Amine-blocked magnetic beads | Surface suitable for conjugation through covalent bonding. Non-surfactant, non-protein-blocked surface. Low non-specific binding. | Conjugation applications, similar to carboxylate-modified beads. |
| Oligo(dT)-coated magnetic beads | Hybridizes with mRNA poly-A tails. High colloidal stability. | mRNA binding applications: mRNA extraction and purification RT-PCR cDNA library construction Subtractive hybridization NGS (RNA sequencing) |
| Streptavidin-coated magnetic beads | Binds biotinylated ligands such as proteins, nucleic acids, and peptides. Covalently bound streptavidin coating. Fast reaction kinetics. Low non-specific binding. High throughput and precision. | Immunoassay and molecular biology applications: Sample preparation and assay development for genomics and proteomics. |
| Streptavidin-blocked magnetic beads | Binds biotinylated ligands such as proteins, nucleic acids, and peptides. Non-surfactant, non-protein-blocked surface. Lower non-specific binding than streptavidin-coated beads via additional blocking of non-specific binding sites. | High-specificity biotin binding applications Molecular and immunodiagnostics NGS library preparation |
| NeutrAvidin™-coated magnetic beads | Binds biotinylated ligands such as proteins, nucleic acids, and peptides. Fast reaction kinetics. Low non-specific binding. High throughput and precision. | Alternative to Streptavidin in immunoassay and molecular biology applications: Sample preparation and assay development for genomics and proteomics. |
| Protein A/G magnetic beads | Binds IgA and IgG proteins Coating based on IgA/IgG fusion protein. Broad binding capabilities. | Antibody isolation applications: Affinity purification and pull-down Immunoprecipitation |
| Silica-coated magnetic beads | Reversibly binds nucleic acids based on salt concentration. Monodisperse particles with narrow size ranges of 400 µm or 700 µm. | Applications with low sample amounts Nucleic acid extraction for molecular diagnostics applications such as qPCR. |
| Mag-sepharose | Broad range of ligand options. Porous, providing greater surface area than other magnetic beads. | Convenient alternative to sepharose columns, with protein purification applications including: Affinity purification or capture Immunoprecipitation |
Factors That Influence Quality of Isolated Nucleic Acids
The integrity of pure genomic DNA is significantly affected by various isolation procedures. Mammalian chromosomal DNA is several hundred mega base pairs (mbp) in size that is large as compared to a few kilobasepairs of plasmid DNA that is quickly split as subjected to blunt cell extract mechanical shaving forces that strike DNA untangled from chromatin proteins. Centrifugation-based separation techniques with diligent pipetting allow for the gentle isolation of nucleic acid, resulting in strong nucleic acid integrity.
Nucleic acid purity is an significant factor which researchers evaluate when choosing the method of isolation. Impurities such as carbohydrates , proteins, or organic compounds coelute with Nucleic Acid in suboptimal conditions. For downstream uses, either allows Nucleic Acid insoluble, degrades it or prevents enzyme function. Carryover of these compounds affects spectroscopic readings and may result in underestimation / overestimation of the yield of nucleic acid, along with other considerations such as pH. Market kits shall have percentages of purity within the prescribed range. These are planned to have sufficient purity of the 260/280 nm absorption ratios being ~1.8 for DNA and ~2 for RNA. The 260/230 values are usually in the 2.0–2.2 range. If the ratio is appreciably smaller than predicted, the presence of pollutants that absorb at 230 nm may be suggested. Some organic compounds used in nucleic acid purification have up to 230 nm of absorbance. Those are: EDTA, glucose, phenol, Guanidine HCl; whereas the isothiocyanate of guanidine used for RNA isolation absorbs at 260 nm.
Conclusion
Extractions of nucleic acid based on magnetic beads are well known in the research, clinical, and forensics field. Overall, their prominence is expanding and in the near future, both in the advanced laboratories and in low-resource environments, they will have much greater use. The fact that magnetic beads are not only used for nucleic acid separation but also for cancer diagnosis, imaging and treatment, fuels research in this area. And in the near future we might foresee much better fabrics.
References:
- Kovačević, N. (2016). Magnetic Beads Based Nucleic Acid Purification for Molecular Biology Applications. Sample Preparation Techniques for Soil, Plant, and Animal Samples, 53–67. doi:10.1007/978-1-4939-3185-9_5
- Magbeads 101: A guide to choosing and using magnetic beads

hello ;
thank you for your informtion about this technology ,
but i have question about the buffer used in this technology
can you give this buffer(lysis buffer ,binding buffer ,wanshing buffer …)
thank you
hi,
Thank you for your organization about DNA extraction with magnetic beads,
Could you share about Reference of this information?
Updated. Thank you.